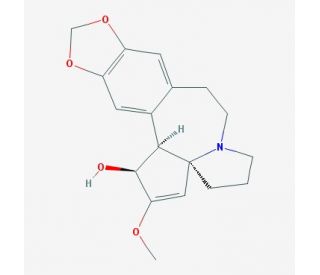
Cephalotaxine (CAS 24316-19-6)
QUICK LINKS
Cephalotaxine is an alkaloid isolated from the leaves and bark of trees belonging to the genus Cephalotaxus. Structurally, cephalotaxine is characterized by its unique bicyclic ring system, which includes a fused dihydroindole and tetrahydroisoquinoline, making it a distinctive member of the alkaloid family. The primary mechanism of action of cephalotaxine involves its interaction with tubulin, the protein that polymerizes to form microtubules, which are essential for cell division and cellular structure maintenance. Cephalotaxine binds to tubulin at a site distinct from that of other well-known tubulin inhibitors, leading to the destabilization of microtubule formation. This disruption prevents the assembly of the microtubule network, which is crucial for the mitotic spindle formation during cell division, thereby arresting cells in the metaphase of mitosis. In research, cephalotaxine has been used extensively to study the dynamics of microtubule assembly and its role in cell cycle regulation. By inhibiting microtubule formation, cephalotaxine serves as a tool to understand the effects of altered microtubule dynamics on cellular processes such as migration, nutrient uptake, and the intracellular transport of organelles and molecules. Additionally, cephalotaxine has been employed in studies exploring the cellular response to mitotic arrest. It aids in the investigation of the spindle assembly checkpoint, a critical cell cycle checkpoint that ensures proper chromosome alignment before segregation. Understanding how cells respond to microtubule disruption can reveal much about the mechanisms that maintain genomic stability and prevent aneuploidy.
Cephalotaxine (CAS 24316-19-6) References
- Formal syntheses of (+/-)-stemonamine and (+/-)-cephalotaxine. | Zhao, YM., et al. 2009. J Org Chem. 74: 3211-3. PMID: 19320459
- Formal synthesis of cephalotaxine. | Zhang, ZW., et al. 2013. J Org Chem. 78: 786-90. PMID: 23214930
- Omacetaxine mepesuccinate in chronic myeloid leukemia. | Al Ustwani, O., et al. 2014. Expert Opin Pharmacother. 15: 2397-405. PMID: 25301179
- Homoharringtonine-induced hyperglycemia. | Sylvester, RK., et al. 1989. J Clin Oncol. 7: 392-5. PMID: 2645387
- Anti-varicella-zoster virus activity of cephalotaxine esters in vitro. | Kim, JE. and Song, YJ. 2019. J Microbiol. 57: 74-79. PMID: 30456755
- Cephalotaxine inhibits Zika infection by impeding viral replication and stability. | Lai, ZZ., et al. 2020. Biochem Biophys Res Commun. 522: 1052-1058. PMID: 31818462
- Cephalotaxine Inhibits the Survival of Leukemia Cells by Activating Mitochondrial Apoptosis Pathway and Inhibiting Autophagy Flow. | Liu, T., et al. 2021. Molecules. 26: PMID: 34070111
- Effective therapy for AML with RUNX1 mutation by cotreatment with inhibitors of protein translation and BCL2. | Mill, CP., et al. 2022. Blood. 139: 907-921. PMID: 34601571
- Inhibition of the CDK2 and Cyclin A complex leads to autophagic degradation of CDK2 in cancer cells. | Zhang, J., et al. 2022. Nat Commun. 13: 2835. PMID: 35595767
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cephalotaxine, 5 mg | sc-211054 | 5 mg | $180.00 |
