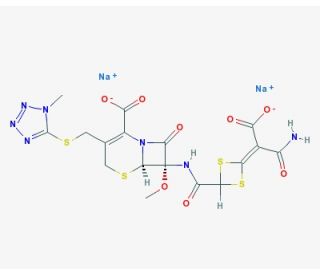
Cefotetan disodium (CAS 74356-00-6)
QUICK LINKS
Cefotetan disodium, a second-generation cephamycin antibiotic, has garnered attention in scientific research due to its potent antibacterial properties and unique mechanism of action. It exhibits broad-spectrum activity against both Gram-positive and Gram-negative bacteria, making it a valuable tool in studying bacterial resistance mechanisms and exploring novel approaches. Research has delved into elucidating the molecular basis of its antimicrobial activity, revealing its ability to inhibit bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs). Furthermore, studies have investigated its efficacy in combating multidrug-resistant bacterial strains, shedding light on potential strategies to overcome antibiotic resistance. Additionally, cefotetan disodium has been utilized in microbiological research to investigate bacterial cell wall dynamics and the mechanisms underlying bacterial cell death. Its role as a research tool extends to exploring bacterial physiology, pathogenesis, and evolutionary adaptations, providing insights into fundamental aspects of microbial biology. Overall, cefotetan disodium stands as a valuable resource in scientific investigations aimed at understanding bacterial biology, antibiotic resistance mechanisms, and developing novel antimicrobial strategies.
Cefotetan disodium (CAS 74356-00-6) References
- Cefotetan disodium-induced hemolytic anemia. | Wagner, BK., et al. 1992. Ann Pharmacother. 26: 199-200. PMID: 1554932
- Low-dose carbon monoxide inhibits progressive chronic allograft nephropathy and restores renal allograft function. | Nakao, A., et al. 2009. Am J Physiol Renal Physiol. 297: F19-26. PMID: 19369289
- A simple and sensitive liquid chromatographic technique for the determination of cefotetan disodium in human plasma and its application in a pharmacokinetic study. | Liu, Y., et al. 2012. J Huazhong Univ Sci Technolog Med Sci. 32: 779-784. PMID: 23073813
- Palonosetron hydrochloride compatiblity and stability with three B-lactam anitbiotic during simulated y-site adminstration. | Ben, M., et al. 2007. Int J Pharm Compd. 11: 520-4. PMID: 23994813
- Effects of cefotetan disodium, cefoxitin, cefazolin, and cefotaxime in vitro on polymorphonuclear leukocytes from patients with leukopenia and severe pelvic inflammatory disease. | Ford, LC., et al. 1988. Am J Obstet Gynecol. 158: 744-5. PMID: 3162655
- Interference of cefmetazole sodium and cefotetan disodium with urine-glucose testing systems. | Stanfield, JA. and DiPiro, JT. 1988. Am J Hosp Pharm. 45: 625-6. PMID: 3163470
- The development of hypoprothrombinaemia following antibiotic therapy in malnourished patients with low serum vitamin K1 levels. | Cohen, H., et al. 1988. Br J Haematol. 68: 63-6. PMID: 3422816
- An embryotoxicity study in Macaca fascicularis with cefotetan disodium (a cephamycin antibiotic). | Allen, DG., et al. 1982. Toxicol Lett. 11: 43-7. PMID: 6953621
- Incompatibility of cefotetan disodium and promethazine hydrochloride. | Erickson, SH. and Ulici, D. 1995. Am J Health Syst Pharm. 52: 1347. PMID: 7656125
- Palonosetron Hydrochloride Compatibility and Stability with Three Beta-Lactam Antibiotics During Simulated Y-Site Administration | Ben, M., et al. 2007. International Journal of Pharmaceutical Compounding. 11(6): 520.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cefotetan disodium, 100 mg | sc-357307 | 100 mg | $76.00 |
