Cefotaxime (CAS 63527-52-6)
See product citations (1)
QUICK LINKS
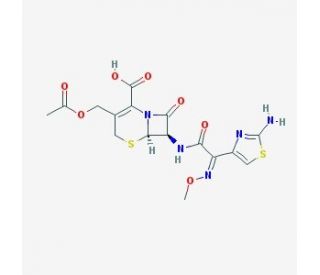
Cefotaxime, a third-generation cephalosporin antibiotic, has garnered significant attention in scientific research owing to its potent antibacterial properties. Its mechanism of action involves inhibiting bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs), particularly PBP-3 in Gram-negative bacteria. This leads to impaired peptidoglycan cross-linking, ultimately causing bacterial cell lysis and death. In research, cefotaxime has been instrumental in microbiology studies, serving as a crucial tool for bacterial culture and antibiotic susceptibility testing. Additionally, its efficacy against a wide range of Gram-negative pathogens, including Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa, has made it indispensable for investigating bacterial resistance mechanisms and developing novel strategies. Furthermore, cefotaxime′s ability to penetrate the blood-brain barrier has facilitated its use in neuroscience research, particularly in studies exploring the role of bacterial infections in neurological disorders. Overall, cefotaxime continues to be a cornerstone in scientific investigations aimed at understanding bacterial physiology, pathogenesis, and antimicrobial resistance, providing invaluable insights into combating infectious diseases in various research domains.
Cefotaxime (CAS 63527-52-6) References
- Oral versus intravenous empirical antimicrobial therapy for fever in patients with granulocytopenia who are receiving cancer chemotherapy. International Antimicrobial Therapy Cooperative Group of the European Organization for Research and Treatment of Cancer. | Kern, WV., et al. 1999. N Engl J Med. 341: 312-8. PMID: 10423465
- [Antibiotic prophylaxis of postoperative complications in surgical treatment of pulmonary, tracheal and mediastinal tumors]. | Petrova, MV., et al. 2001. Anesteziol Reanimatol. 58-60. PMID: 11757305
- Cefotaxime is extensively used for surgical prophylaxis. | Sader, HS. and Jones, RN. 1992. Am J Surg. 164: 28S-38S. PMID: 1443358
- Cefotaxime in urinary tract infections. | Naber, KG. 1989. Infection. 17: 425-8. PMID: 2693361
- Cefotaxime Exposure Selects Mutations within the CA-Domain of envZ Which Promote Antibiotic Resistance but Repress Biofilm Formation in Salmonella. | Trampari, E., et al. 2022. Microbiol Spectr. 10: e0214521. PMID: 35475640
- Elimination of cefotaxime using polysulfone and polyacrylonitrile-derived filters: An in vitro assessment. | Le Ven, J., et al. 2023. Int J Artif Organs. 46: 113-119. PMID: 36583520
- The effects of switching from ceftriaxone to cefotaxime on the occurrence of third-generation cephalosporin-resistant Enterobacterales: A stepped-wedge cluster randomized trial. | Bouiller, K., et al. 2024. Infect Dis Now. 54: 104806. PMID: 37838305
- Mechanism of action, antimicrobial activity, pharmacology, adverse effects, and clinical efficacy of cefotaxime. | LeFrock, JL., et al. 1982. Pharmacotherapy. 2: 174-84. PMID: 6302641
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cefotaxime, 1 g | sc-353131 | 1 g | $292.00 | |||
Cefotaxime, 5 g | sc-353131A | 5 g | $938.00 |
