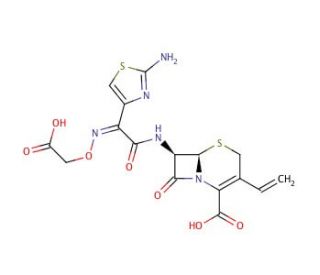
Cefixime (CAS 79350-37-1)
QUICK LINKS
Cefixime is a broad-spectrum antibiotic that inhibits bacterial cell wall synthesis by binding to penicillin-binding proteins. This binding prevents the final transpeptidation step of peptidoglycan synthesis in bacterial cell walls, leading to cell lysis and death. Cefixime targets the enzymes involved in the cross-linking of the peptidoglycan polymers, disrupting the structural integrity of the bacterial cell wall. This disruption ultimately leads to the inhibition of bacterial growth and reproduction. Cefixime exhibits bactericidal activity against a wide range of gram-negative and gram-positive bacteria, making it useful for studying bacterial cell wall synthesis and the mechanisms of antibiotic resistance. Its mechanism of action provides insight into the molecular interactions involved in bacterial cell wall synthesis and the development of potential strategies to combat antibiotic resistance.
Cefixime (CAS 79350-37-1) References
- Cefixime in urinary tract infections with special reference to pediatrics: overview. | Fanos, V. and Cataldi, L. 2001. J Chemother. 13: 112-7. PMID: 11330356
- Cefixime-induced oculogyric crisis. | Bayram, E., et al. 2012. Pediatr Emerg Care. 28: 55-6. PMID: 22217888
- Clinical Efficacy of Cefixime for the Treatment of Early Syphilis. | Stafylis, C., et al. 2021. Clin Infect Dis. 73: 907-910. PMID: 33640982
- Cefixime and cefixime-clavulanate for screening and confirmation of extended-spectrum beta-lactamases in Escherichia coli. | Al-Tamimi, M., et al. 2022. Ann Clin Microbiol Antimicrob. 21: 20. PMID: 35599329
- Randomized controlled trial of the relative efficacy of high-dose intravenous ceftriaxone and oral cefixime combined with doxycycline for the treatment of Chlamydia trachomatis and Neisseria gonorrhoeae co-infection. | Nguyen, PTT., et al. 2022. BMC Infect Dis. 22: 607. PMID: 35810277
- Molecular docking, molecular dynamics simulations and in vitro screening reveal cefixime and ceftriaxone as GSK3β covalent inhibitors. | Nassar, H., et al. 2023. RSC Adv. 13: 11278-11290. PMID: 37057264
- Clinical pharmacokinetics of cefixime: a systematic review. | Ajmal, M., et al. 2023. Xenobiotica. 53: 149-162. PMID: 37216497
- In situ preparation of MOF-199 into the carrageenan-grafted-polyacrylamide@Fe3O4 matrix for enhanced adsorption of levofloxacin and cefixime antibiotics from water. | Salehi, MM., et al. 2023. Environ Res. 233: 116466. PMID: 37348634
- Cefixime-tellurite-deoxycholate tryptic soy broth (CTD-TSB), a selective enrichment medium, for enhancing isolation of Escherichia albertii from wild raccoon fecal samples. | Xu, B., et al. 2023. J Appl Microbiol. 134: PMID: 37370227
- Biosynthesis approach of copper nanoparticles, physicochemical characterization, cefixime wastewater treatment, and antibacterial activities. | Hassan, E., et al. 2023. BMC Chem. 17: 71. PMID: 37424027
- Antimicrobial susceptibilities and genomic epidemiology of Neisseria gonorrhoeae in Stockholm, Sweden. | Vestberg, N., et al. 2023. Eur J Clin Microbiol Infect Dis.. PMID: 37442885
- Antimicrobial Resistance Profiling and Genome Analysis of the penA-60.001 Neisseria gonorrhoeae Clinical Isolates in China in 2021. | Tang, Y., et al. 2023. J Infect Dis.. PMID: 37462263
- Cefixime. A review of its therapeutic efficacy in lower respiratory tract infections. | Markham, A. and Brogden, RN. 1995. Drugs. 49: 1007-22. PMID: 7641600
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cefixime, 10 mg | sc-217857 | 10 mg | $150.00 |
