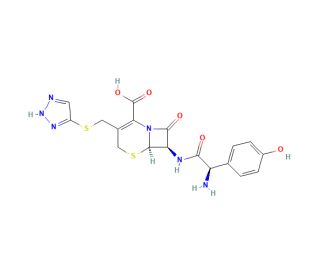
Cefatrizine (CAS 51627-14-6)
QUICK LINKS
Cefatrizine is a semi-synthetic second-generation cephalosporin, primarily explored for its biochemical properties and mechanisms of action in bacterial cell processes. It targets the synthesis of the bacterial cell wall, specifically by interacting with penicillin-binding proteins (PBPs). This interaction disrupts the transpeptidation process of peptidoglycan assembly, which is crucial for maintaining cell wall integrity. In scientific research, cefatrizine′s effects on PBPs have been utilized to understand the structural dynamics and enzymatic activities involved in cell wall assembly. By inhibiting these critical processes, cefatrizine serves as a tool for studying the biochemical pathways essential to bacterial cell survival and growth. The research on cefatrizine also extends to examining how certain bacterial strains can alter their cell wall structure and function in response to external chemical pressures. This has implications for studying broader biological phenomena, such as cellular adaptation to environmental changes.
Cefatrizine (CAS 51627-14-6) References
- Susceptibility of Haemophilus influenzae type b to cefatrizine, ampicillin, and chloramphenicol. | Brotherton, T., et al. 1976. Antimicrob Agents Chemother. 10: 322-4. PMID: 1086634
- Simultaneous determination of cefatrizine and clavulanic acid in dog plasma by HPLC. | Choi, HG., et al. 2004. J Pharm Biomed Anal. 35: 221-31. PMID: 15030898
- Integrative bioinformatics and proteomics-based discovery of an eEF2K inhibitor (cefatrizine) with ER stress modulation in breast cancer cells. | Yao, Z., et al. 2016. Mol Biosyst. 12: 729-36. PMID: 26776155
- Susceptibility of clinical isolates of gram-positive and gram-negative organisms to cefatrizine. | Carlone, NA., et al. 1985. Drugs Exp Clin Res. 11: 447-51. PMID: 3836867
- [Studies on sub-MIC activities of beta-lactam antibiotics and aminoglycoside antibiotics against clinically isolated strain]. | Takahashi, M., et al. 1985. Jpn J Antibiot. 38: 3497-504. PMID: 3938820
- Comparison of in vitro antibacterial activity of four oral cephems: cephalexin, cefaclor, cefatrizine and cefadroxil. | Luh, KT., et al. 1984. Zhonghua Min Guo Wei Sheng Wu Ji Mian Yi Xue Za Zhi. 17: 11-8. PMID: 6378550
- Laboratory studies with cefatrizine (SK + F 60771), a new broad-spectrum orally-active cephalosporin. | Actor, P., et al. 1975. J Antibiot (Tokyo). 28: 594-601. PMID: 808525
- Activity of four cephalosporin antibiotics in vitro against bovine udder pathogens and pathogenic bacteria isolated from newborn calves. | Ziv, G. 1976. Antimicrob Agents Chemother. 9: 418-21. PMID: 816249
- In vitro activities of five oral cephalosporins against aerobic pathogenic bacteria. | Shadomy, S., et al. 1977. Antimicrob Agents Chemother. 12: 609-13. PMID: 921258
- In vitro evaluation of the new oral cephalosporin cefatrizine: comparison with other cephalosporins. | Blackwell, CC., et al. 1976. Antimicrob Agents Chemother. 10: 288-92. PMID: 984771
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cefatrizine, 100 mg | sc-504726 | 100 mg | $423.00 |
