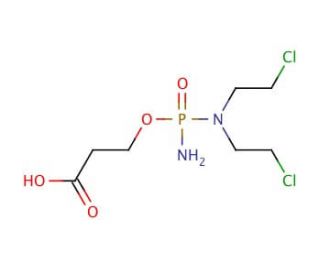
Carboxyphosphamide (CAS 22788-18-7)
See product citations (1)
QUICK LINKS
Carboxyphosphamide is a chemical compound that functions as an alkylating agent in research and development. Its mechanism of action involves the covalent binding of its active metabolites to DNA, leading to the formation of interstrand and intrastrand cross-links. This process interferes with DNA replication and transcription, ultimately inducing cell death. Carboxyphosphamide′s ability to induce DNA damage and inhibit cell proliferation makes it a useful tool for studying the molecular mechanisms of DNA repair and the cellular response to genotoxic stress. Its role in experimental contexts involves elucidating the pathways involved in DNA damage response. By understanding its mode of action at the molecular level, researchers can gain insights into the fundamental processes underlying cell cycle regulation and DNA repair mechanisms.
Carboxyphosphamide (CAS 22788-18-7) References
- Carboxyphosphamide: NMR studies of its stability and cell membrane permeability. | Ludeman, SM., et al. 1992. Drug Metab Dispos. 20: 337-8. PMID: 1352232
- Cyclophosphamide metabolism in children with non-Hodgkin's lymphoma. | Yule, SM., et al. 2004. Clin Cancer Res. 10: 455-60. PMID: 14760065
- The effect of cimetidine on cyclophosphamide metabolism in rabbits. | Anthony, LB., et al. 1990. Cancer Chemother Pharmacol. 27: 125-30. PMID: 2249327
- Selective ALDH3A1 inhibition by benzimidazole analogues increase mafosfamide sensitivity in cancer cells. | Parajuli, B., et al. 2014. J Med Chem. 57: 449-61. PMID: 24387105
- Kinetic characterization of the catalysis of 'activated' cyclophosphamide (4-hydroxycyclophosphamide/aldophosphamide) oxidation to carboxyphosphamide by mouse hepatic aldehyde dehydrogenases. | Manthey, CL. and Sladek, NE. 1988. Biochem Pharmacol. 37: 2781-90. PMID: 3395357
- Phenotypically deficient urinary elimination of carboxyphosphamide after cyclophosphamide administration to cancer patients. | Hadidi, AH., et al. 1988. Cancer Res. 48: 5167-71. PMID: 3409242
- Combined thin-layer chromatography-photography-densitometry for the quantitation of cyclophosphamide and its four principal urinary metabolites. | Hadidi, AH. and Idle, JR. 1988. J Chromatogr. 427: 121-30. PMID: 3410892
- 3D-QSAR and scaffold hopping based designing of benzo[d]ox-azol-2(3H)-one and 2-oxazolo[4,5-b]pyridin-2(3H)-one derivatives as selective aldehyde dehydrogenase 1A1 inhibitors: Synthesis and biological evaluation. | Verma, H., et al. 2022. Arch Pharm (Weinheim). 355: e2200108. PMID: 35618489
- Metabolism of high doses of cyclophosphamide. | Milsted, RA. and Jarman, M. 1982. Cancer Chemother Pharmacol. 8: 311-3. PMID: 7127662
- Inhibition by cyanamide of 4-hydroxycyclophosphamide/aldophosphamide oxidation to carboxyphosphamide. | Domeyer, BE. and Sladek, NE. 1981. Biochem Pharmacol. 30: 2065-73. PMID: 7295327
- Cyclophosphamide metabolism in children. | Yule, SM., et al. 1995. Cancer Res. 55: 803-9. PMID: 7850793
- Dose escalation of cyclophosphamide in patients with breast cancer: consequences for pharmacokinetics and metabolism. | Busse, D., et al. 1997. J Clin Oncol. 15: 1885-96. PMID: 9164199
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Carboxyphosphamide, 5 mg | sc-207411 | 5 mg | $273.00 |
