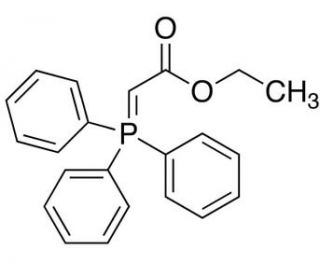
Molecular structure of (Carbethoxymethylene)triphenylphosphorane, CAS Number: 1099-45-2
(Carbethoxymethylene)triphenylphosphorane (CAS 1099-45-2)
Alternate Names:
(Ethoxycarbonylmethylene)triphenylphosphorane; Ethyl (triphenylphosphoranylidene)acetate
Application:
(Carbethoxymethylene)triphenylphosphorane is a Wittig reagent and cholinesterase inhibitor
CAS Number:
1099-45-2
Purity:
≥98%
Molecular Weight:
348.37
Molecular Formula:
C22H21O2P
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
(Carbethoxymethylene)triphenylphosphorane is a phosphorane compound for Wittig olefinations. (Carbethoxymethylene)triphenylphosphorane has been utilized towards the synthesis of isosorbide-based cholinesterase inhibitors.
(Carbethoxymethylene)triphenylphosphorane (CAS 1099-45-2) References
- Isosorbide-based cholinesterase inhibitors; replacement of 5-ester groups leading to increased stability. | Dillon, GP., et al. 2010. Bioorg Med Chem. 18: 1045-53. PMID: 20093035
- Intramolecular azide to alkene cycloadditions for the construction of pyrrolobenzodiazepines and azetidino-benzodiazepines. | Hemming, K., et al. 2014. Molecules. 19: 16737-56. PMID: 25329868
- α-Tetrasubstituted Aldehydes through Electronic and Strain-Controlled Branch-Selective Stereoselective Hydroformylation. | Eshon, J., et al. 2018. J Org Chem. 83: 10207-10220. PMID: 30070109
- Modular Synthesis and Biological Investigation of 5-Hydroxymethyl Dibenzyl Butyrolactones and Related Lignans. | Davidson, SJ., et al. 2018. Molecules. 23: PMID: 30467285
- Development of near-infrared firefly luciferin analogue reacted with wild-type and mutant luciferases. | Kitada, N., et al. 2020. Chirality. 32: 922-931. PMID: 32367573
- Inhibitory Properties of Aldehydes and Related Compounds against Phytophthora infestans-Identification of a New Lead. | Mackrill, JJ., et al. 2020. Pathogens. 9: PMID: 32645837
- Discovery and Optimization of 7-Alkylidenyltetrahydroindazole-Based Acylsulfonamide EP3 Antagonists. | Zhu, B., et al. 2022. ACS Med Chem Lett. 13: 111-117. PMID: 35059130
- Comprehensive Review of Recent Advances in Chiral A-Ring Flavonoid Containing Compounds: Structure, Bioactivities, and Synthesis. | Zhang, C., et al. 2023. Molecules. 28: PMID: 36615559
- Anti-Microbial Activity of Aliphatic Alcohols from Chinese Black Cardamom (Amomum tsao-ko) against Mycobacterium tuberculosis H37Rv. | Lee, SY., et al. 2022. Plants (Basel). 12: PMID: 36616162
- Synthesis and Anti-Proliferative Evaluation of Arctigenin Analogues with C-9' Derivatisation. | Paulin, EK., et al. 2023. Int J Mol Sci. 24: PMID: 36674683
- A Redox-Relay Heck Approach to Substituted Tetrahydrofurans. | Byrne, TJM., et al. 2023. Org Lett. 25: 2361-2365. PMID: 36988968
- Synthesis of Gamma-Butyrolactone Hormones Enables Understanding of Natural Product Induction. | Wilbanks, LE., et al. 2023. ACS Chem Biol. 18: 1624-1631. PMID: 37338162
- Flow Chemistry for Synthesis of 2-(C-Glycosyl)acetates from Pyranoses via Tandem Wittig and Michael Reactions. | Bennett, JJ. and Murphy, PV. 2024. Org Process Res Dev. 28: 1848-1859. PMID: 38783857
Inhibitor of:
AChE, and BChE.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(Carbethoxymethylene)triphenylphosphorane, 50 g | sc-207403 | 50 g | $175.00 |
