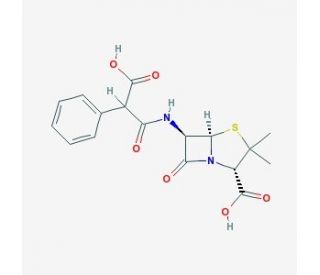
Carbenicillin solution
QUICK LINKS
Carbenicillin, a β-lactam antibiotic derived from penicillin, serves as a vital tool in scientific research, particularly in the field of molecular biology and microbiology. Mechanistically, carbenicillin exerts its antimicrobial activity by inhibiting bacterial cell wall synthesis through irreversible binding to penicillin-binding proteins (PBPs), enzymes involved in peptidoglycan cross-linking. This inhibition leads to the disruption of bacterial cell wall integrity, ultimately resulting in cell lysis and death. Moreover, carbenicillin is widely used as a selective agent in molecular biology applications due to its ability to selectively kill bacteria carrying plasmids encoding resistance genes, such as ampicillin resistance genes (e.g., blaTEM). This property enables the isolation and propagation of recombinant DNA molecules in bacterial hosts, facilitating the cloning and expression of foreign genes. Furthermore, carbenicillin is employed in microbiological research to study bacterial genetics, antibiotic resistance mechanisms, and microbial ecology. Researchers utilize carbenicillin-containing growth media to culture and select bacteria harboring specific genetic traits or mutations, aiding in the investigation of bacterial physiology, gene regulation, and microbial interactions. Additionally, carbenicillin has been utilized in various studies investigating bacterial pathogenesis, biofilm formation, and antibiotic susceptibility patterns. Overall, the versatile applications of carbenicillin in scientific research contribute significantly to advancing our understanding of bacterial biology, genetic engineering, and antibiotic resistance mechanisms.
Carbenicillin solution References
- Construction of gene expression system in hop (Humulus lupulus) lupulin gland using valerophenone synthase promoter. | Okada, Y., et al. 2003. J Plant Physiol. 160: 1101-8. PMID: 14593812
- Complexation of beta-lactam antibiotic drug carbenicillin to bovine serum albumin: energetics and conformational studies. | Thoppil, AA., et al. 2008. Biopolymers. 89: 831-40. PMID: 18488985
- Determination of acyl homoserine lactone and tetramic acid concentrations in biological samples. | Lowery, CA., et al. 2011. Methods Mol Biol. 692: 101-11. PMID: 21031307
- Differential scanning calorimetry to quantify the stability of protein cages. | Zhang, Y. and Ardejani, MS. 2015. Methods Mol Biol. 1252: 101-13. PMID: 25358777
- Purification of bacterial RNA polymerase: tools and protocols. | Svetlov, V. and Artsimovitch, I. 2015. Methods Mol Biol. 1276: 13-29. PMID: 25665556
- In vitro susceptibility of Pseudomonas aeruginosa to carbenicillin and the combination of carbenicillin and gentamicin. | Phair, JP., et al. 1969. Appl Microbiol. 18: 303-6. PMID: 4984762
- Hydroxylamine technique for in vitro prevention of penicillin inactivation of tobramycin. | Falkowski, AJ. and Creger, RJ. 1984. Antimicrob Agents Chemother. 26: 643-6. PMID: 6393865
- Root formation in micropropagated shoots of Sequoia sempervirens using Agrobacterium | Snježana Mihaljević a, Vesna Katavić b, Sibila Jelaska a c. 1999. Plant Science. 141: 73-80.
- Transformation of androgenic-derived Festulolium plants (Lolium perenne L. × Festuca pratensis Huds.) by Agrobacterium tumefaciens | Yang-Dong Guo, Hisano Hisano, Yoshiya Shimamoto & Toshihiko Yamada. 2009. Plant Cell, Tissue and Organ Culture (PCTOC). 96: 219–227.
- Kinetics and Mechanism of Oxidation of Carbenicillin by Copper (III) Periodate Complex in Aqueous Alkaline Medium | Yuv Raj Sahu, Parashuram Mishra. 2020. Journal of Chemistry. 4060984.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Carbenicillin solution, 1 ml | sc-278810 | 1 ml | $437.00 |
