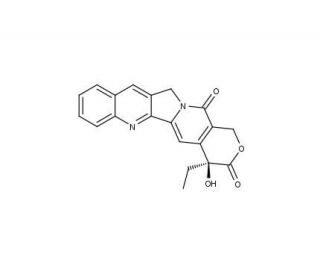
Camptothecin 의 분자 구조, CAS 번호: 7689-03-4
Camptothecin (CAS 7689-03-4)
인용논문 보기 (21)
대체 이름:
(S)-(+)-Camptothecin
적용:
Camptothecin 는 항종양 활성을 나타내는 가역적 미토콘드리아 토포 I 억제제입니다.
CAS 등록번호:
7689-03-4
순도:
≥98%
분자량:
348.4
분자식:
C20H16N2O4
추가 정보:
운송용 위험물로 분류되어 추가 배송료가 부과될 수 있습니다.
연구용으로만 사용가능합니다. 진단이나 치료용으로 사용불가합니다.
* 참조분석증명서대량의 측정 데이터(함수량포함).
빠른 링크
주문정보
논문정보
설명
기술정보
안전정보
SDS 및 분석 증명서
캄토테신은 원래 중국 나무 캄토테카 아쿠미나타에서 발견되는 알칼로이드 화합물이다. 연구 맥락에서, 캄토테신은 DNA 복제와 전사에 중요한 효소인 DNA 토포이소머라제 I을 억제하는 능력으로 평가된다. 토포이소머라제 I이 생성하는 이중 가닥 DNA의 일시적인 분열을 안정화함으로써, 캄토테신은 DNA 손상과 궁극적으로 세포 사멸을 유도하는데, 이는 특히 세포 증식과 생존에 관한 연구와 관련이 있다. 이러한 특성은 암세포가 증식하고 생존하기 위해 사용하는 메커니즘 연구뿐만 아니라 통제된 세포 사멸을 유도하기 위한 잠재적인 전략을 탐구하는 데 초점이 되었다. DNA 손상 반응 경로를 통해 세포 사멸을 유도하는 그것의 역할은 세포 생존과 죽음 사이의 복잡한 균형을 이해하는 데 더욱 기여한다. 또한 캄토테신에 대한 연구는 분자 생물학 분야로 확장되어 DNA 복제의 역학과 유전독성 스트레스에 대한 세포 반응에 대한 견해를 제공한다.
Camptothecin (CAS 7689-03-4) 참고자료
- 정맥 주사한 캠토테신 고체 지질 나노입자의 생쥐 체내 분포 및 뇌에 대한 표적 효과. | Yang, SC., et al. 1999. J Control Release. 59: 299-307. PMID: 10332062
- 리포솜 에어로졸로 전달한 후 또는 마우스에 근육 주사 후 캠토테신을 분포시킵니다. | Koshkina, NV., et al. 1999. Cancer Chemother Pharmacol. 44: 187-92. PMID: 10453719
- 캠토테신은 RAW 264.7 대식세포에서 산화질소 생합성을 억제합니다. | Chiou, WF., et al. 2001. Life Sci. 69: 625-35. PMID: 11476184
- 캠토테신과 그 유사체: 화학 요법 잠재력에 대한 검토. | Sriram, D., et al. 2005. Nat Prod Res. 19: 393-412. PMID: 15938148
- DNA-토포이소머라제 I 공유 결합체의 비가역적 트래핑. 캠토테신 결합 부위의 친화성 라벨링. | Hertzberg, RP., et al. 1990. J Biol Chem. 265: 19287-95. PMID: 2172250
- 마우스에서 캠토테신 유사체 CPT-11의 대사 및 약동학. | Kaneda, N., et al. 1990. Cancer Res. 50: 1715-20. PMID: 2306725
- 노타포디테스 님모니아나에서 분리된 알터나리아(Alternaria sp.)에서 캠토테신의 지속 가능한 생산. | Mohinudeen, IAHK., et al. 2021. Sci Rep. 11: 1478. PMID: 33446714
- 캠토테신의 항종양 활성, 작용 기전 및 세포 주기 효과에 대한 연구. | Gallo, RC., et al. 1971. J Natl Cancer Inst. 46: 789-95. PMID: 4995657
- 캠토테신 유도체에 의한 진핵생물 DNA 토포이소머라제 I 절단 가능 복합체의 차등적 안정화. | Tanizawa, A., et al. 1995. Biochemistry. 34: 7200-6. PMID: 7766631
- 항암제로서 캠토테신 유사체의 현재 상태. | Slichenmyer, WJ., et al. 1993. J Natl Cancer Inst. 85: 271-91. PMID: 8381186
- 항암제 캠토테신은 신장을 억제하지만 RNA 중합 효소 II 전사의 시작을 자극합니다. | Ljungman, M. and Hanawalt, PC. 1996. Carcinogenesis. 17: 31-5. PMID: 8565133
- 화학요법제 CPT-11은 세포질에 세포사멸 개시 인자의 새로운 발현을 유도합니다. | Suzuki, A. and Kato, M. 1996. Exp Cell Res. 227: 154-9. PMID: 8806462
- p53-null 인간 백혈병 HL60 세포 및 분리된 핵에서 캠토테신에 의한 세포 사멸: 프로테아제 억제제 Z-VAD-fmk 및 디클로로이소쿠마린의 효과는 카스파제와 세린 프로테아제 모두의 관여를 시사합니다. | Shimizu, T. and Pommier, Y. 1997. Leukemia. 11: 1238-44. PMID: 9264376
- 진핵생물 DNA 토포이소머라제 I과 이 효소를 표적으로 하는 약물의 작용 메커니즘. | Pommier, Y., et al. 1998. Biochim Biophys Acta. 1400: 83-105. PMID: 9748515
의 억제제:
(eukaryotic translation initiation factor) eIF, (eukaryotic translation initiation factor) eIF1, (eukaryotic translation initiation factor) eIF2, (eukaryotic translation initiation factor) eIF3, (eukaryotic translation initiation factor) eIF4, (eukaryotic translation initiation factor) eIF5, α-Adaptin 2, α-Dystrobrevin, α3e Tubulin, α8 Tubulin, β-SNAP, 2410076I21Rik, 2610002J02Rik, 4632434I11Rik, 4921530L21Rik, 4930432K21Rik, 4930469G21Rik, 4930547C10Rik, 4931408C20Rik, 6030422M02Rik, 9130206N08Rik, A630018P17Rik, A630023A22Rik, ACD, ACTBL2, ACTR5, ADAM1, ADAT2, Adducin α, ADSL, AGAP3, AI481877, AK4, Alix, ALMS1, ALS, ANKRD12, ANKRD13D, ANKRD18A, ANKRD32, ANKRD54, Annexin A13, Annexin V, Aprataxin, ARD1B, arfaptin 1, ARGLU1, ARL8A, ARMC1, ARMC3, ASAH2C, ASB-17, ATAD5, ATP5L, ATP5L2, ATP9BL, ATPBD3, ATXN3L, ATXN7L3, B930041F14Rik, BAFL, Barx1, BC005561, BC049762, BFZB, BJ-TSA-9, Blood Group N antigen, BMAL2, BMCP1, BOLA1, BRCC3, BRE, Brn-3c, BRP44, BTBD11, BTEB3, BTF3L2, BTF3L4, BTF3L4P, BUD13, C10orf28, C10orf50, C10orf51, C10orf75, C11orf82, C11orf91, C13orf30, C14orf124, C14orf72, C15orf42, C16orf62, C17orf39_4933439F18Rik, C17orf53, C18orf18, C19orf57, C1D, C1orf114, C1orf127, C1orf55, C20orf112, C20orf43, C2orf49_AI597479, C3orf24, C4BPα, C5orf21, C6orf134, C6orf167, C6orf48, C8orf13, C8orf45, C9orf142, CACNG6, CALML6, calsyntenin-3, CARF, CARP-1, CAS4, CCDC4, CCDC57, CCDC66, CCDC79, CCDC98, CCR-9, Cdc45, CDRT15L, CELF5, CENP-B, CENP-L, CENP-S, CHD (chromodomain helicase), CHD7, CHMP1A, Chondrolectin, CIZ1, claudin-20, CMPK, CMTM2a, CMTM2b, Cnbp2, COH1, COL12A, COL9A2, colostrinin, CPEB, CRALBPL, Creatininase, CRISP-10, CrxOS, cryptdin 6, CSDA, CSHL1, Csprs, CST9L, CstF-64T, CstF-77, CTAGE5, CTGLF3, CUL-5, Cux2, CWF19L1, CWF19L2, CXorf39, CXorf57_D330045A20Rik, cyclin O, D930020E02Rik, DBR1, DCAR, Dclre1c, DDX16, DDX24, DDX30, DDX31, DDX35, DDX38, DDX3L, DDX42, DDX52, DDX54, DEAH-box RNA-dependent NTPase (DHX), Dematin, DFF-45, Dim1, DinB, Diversin, DNA Ligase I, DNA Ligase III, DNA pol δ 2, DNA pol δ cat, DNA pol ε B, DNA pol ε cat, DNA pol ε ss, DNA pol λ, DNA pol μ, DNA pol ν, DNA pol ζ, DNA pol 4, DNA2L, DNAHC7B, DND1, DYDC1, Dynlt1e, Dynlt1f, E130309D02Rik, E430018J23Rik, Ear12, Ear7, EG210583, EG210853, EG232801, EG245174, EG245263, EG384589, EG435970, EG546272, EG546282, EG625591, EG628359, eIF1AY, eIF2Bδ, eIF2C2, eIF3M, eIF4B, eIF5A1, ELAC1, Elongin B, ENSMUSG00000073257, ERI-1, ES31, ESCO2, ETAA16, EXDL1, EXTL2, F730047E07Rik, Factor XIII B, FAM108A1, FAM131A, FAM139A, FAM186A, FAM64A, FAST, FER1L6, Fhit, FIGNL2, Filensin, FIV p27, FLJ20433, FLJ43860, FLRT3, FOXI1, FTF, gametogenetin, GAR1, GCNF, Geminin, GFRP, GGNBP1, Ggnbp2, Gm22, GNL2, GOLGA8B, GPSN2, GRINL1A, GS27, GTSE-1, Gucy2d, Gvin1, GW182, H1FOO, H2-M10.4, H2-M2, H2-Q2, H2-T24, H2AL2, H2BFA, Hairy, HAP1A, hCAP-D2, hCG_2015138, hCG_2024596, HcRed, hDcp1a, hDcp2, HEATR5A, HEI10, HEL308, HELIC1, HELIC2, Hfm1, Histatin 1, Histone cluster 1 H2AF, Histone cluster 2 H2BF, Histone cluster 2 H3C, Histone cluster 2 H3D, Histone cluster 2 H4A, Histone cluster 2 H4B, Histone cluster 3 H2A, Histone cluster 3 H2ba, HLA-3, HLTF, HMG-1L1, HMG-I/HMG-Y, HMG20A, HMGI-C, hMLH3, hMSH3, hnRNP A0, hnRNP B1, hnRNP H, hnRNP H′/hnRNP H2, hnRNP M, HNRNPA1L2, Hop2, HPA1, HPR, HPV 16-L1, hSNF2H, HSPC144, HSV-2 gB, hunchback, I-SceI, I830077J02Rik, IFITM3, IFITM6, IFT43, IGHMBP2, Importin-8, Ini1, INO80B, INO80E, INOC1, INTS2, INTS5, IQCF4, ITPase, Jaz, JK-1, Junctophilin-1, Keratin 42, Khdc1c, KIAA0460, KIAA1462, KIAA1530, KIN17, KLH-A, Klk1b3, Klra33, KRT222P, KRTAP10-8, KRTAP11-1, KRTAP14, KRTAP16-7, KRTAP9-L3, LBA1, Lce1a2, Lce1l, LCTL, LDH-AL6A, LEMD1, LEO1, LFY, LIN-54, Lipocalin-11, Lipocalin-5, Liprin α1, LOC100039248, LOC100039284, LOC100039560, LOC100039633, LOC100039786, LOC100039830, LOC100039934, LOC100040201, LOC100040214, LOC100040307, LOC100040682, LOC100040839, LOC100040885, LOC100040981, LOC100041019, LOC100041106, LOC100041119, LOC100041179, LOC143666, LOC198437, LOC255130, LOC284395, LOC285423, LOC345630, LOC388436, LOC388820, LOC441258, LOC441461, LOC642361, LOC643770, LOC643862, LOC644277, LOC645967, LOC646762, LOC727967_BOP1, LOC729978, LOC729991, LRRC55, MA1, MAGP-2, MBP-probe, MCCB, MCFD2L, MCM-BP, MCM3AP, MDGA2, Mec3, MEIOB, MFSD11, MGC87042, Mi2-β, Miner2, Mitochondrial Topo I, mKIAA1530, MLZE, MMR2, MMS19, MND1, MOBKL1A, MOCS1, Morc, Mpa2, MPZL1, MREG, MRT-2, MTP18, MUP17, MUP18, MUS81, MYLC2PL, Myosin VI, N10, NC2α, Ncapd3, Ncapg, NELF, NETO2, NF90, NHP2, Nibrin, Nocturnin, NOL55, NOP16, Nop5, Nop58, NOS, NOS2, NOSTRIN, NPAT, NSE4A, NTH1, Oatp-Y, OBFC1, OBFC2A, OBFC2B, OBP-2A, Odf1, OFCC1, Olfr176, Olfr177, Olfr417, Olfr523, Olfr987, Olfr988, Olr619, OR10H2, OR4C45, OR51AA4, OR52I2, OTTMUSG00000010657, OTTMUSG00000010671, OTTMUSG00000011290, OTTMUSG00000016414, OTTMUSG00000016437, OTTMUSG00000016779, OTTMUSG00000016780, p54/nrb, p91, PABPC1L, PACT, PAI-3, pancpin, Pancreasin, PAPSS 1, PC-PLD4, PDE6C, pescadillo, PGBD1, PheRS, PIF1, PIG-B, PIG-Y, PLC-XD1, PLGLB1, PLK4, PMS1, PMS2, POLDIP2, POLR3E, POM121, Pontin 52, POP5, POTE14, POTEJ, PRDM11, PRIM2A, Protamine 1, PRP19, PRR17, Psf1, Psf3, PSG9, PUS1, quiescin Q6, Rab 22A, Rad53, Rad55, Rad59, RAET1G, Ran, RASAL3, Raver1, Raver2, RBM10, RBM16, RBM19, RBM27, RBM32B, RBMX2, RBMXL3, RBMY1F, RBP1, RCC2, RCCD1, RCOR3, RecA, RECA-1, REF2, REPIN1, REPS1, REXO1, RFC, RFC2, RFC3, RFPL3, Rhox2d, Ribosomal Protein L12, Ribosomal Protein L34, Ribosomal Protein S29, Ribosomal Protein S8, Rim4, RIMBP3C, RMI1, RNF170, RPA 34 kDa subunit, RPA16, RPA40, RPAC2, RPL7A, Rpp14, Rpp20, Rpp38, RPS4X, RQCD1, Rrn3, RRP36, RRS1, Rsf-1, RSL1D1, RUFY4, RuvA, RuvC, RWDD2A, RYBP, SA-2, SAS10, scc-112_Pds5a, Schlafen (SLFN) family, SCML4, SCP-3, Sec24D, SEI-1, Separase, Septin 8, SerpinB6, SETX, SFRS2B, Sgs1, SKIV2L2, Slfn2, Slfn3, SLIRP, SLX1A/B_SLX1, Sm, SMC4, SMC5, SMC6, Smt3, SNAT4, SNM1A, SNM1B, Snrp116, SP140L, SPAG11B, Spartan, SPATA12, Spc24, Spc25, SPF30, SPIN90, SPLUNC3, SPNR, Spo11, SPT16, SPTLC3, ss DNA marker, SSB-4, SSNA1, ST8Sia II, STAG3, Statherin, SUN2, Sval3, SWI/SNF-B, Swi1, SWI5, SYCE1, Sycp2l, Syntaxin 18, TAF I p110_TAF I p95, TAF I p48, TAF II p100, TAF5L, TAFII-28, TASK-2, TBL3, TCTE1, TDE1, TDP1, TDP2, TDRD9, Testican-1, TEX11, TEX15, TFEC, TFIIA, TFIIA-β, TFIIA-γ, TFIID, TFIIE-α, TFIIF RAP 74, TFIIH 2C, TFIIH p8, TFIIIB, TFIIIB90, TFIIIC35, TFIIS, THAP2, THAP9, THOC3, THOC4, THTR2, TIAR, Timm8a2, TISP74, TMCO5B, TMEM16D, TMEM33, TMEM40, TMEM45A, TMEM91, TMPRSS7, TMUB1, TNFα-IP 8L1, Tom40, Top3, Topo, Topo I, Topo IIIβ, TOPORS, TP53TG5, TRABD, TRAG-3, TRAP150, TREX-2, Trim12c, TRIM30C, TRIM35, TRIP, Tropomyosin α, Tropomyosin β, TRPD52L3, TSC-22 D4, TSHZ3, TSLP, TSPAN1, TSR1, TTC21A, TTC22, TTI2_BC019943, TUSC2, Twinfilin-1, Txl-2, TYW4, UBF, UBF1, Ubr4, UDG2, UIMC1, UNC5H4, UNC84A, UPIb, V1RH17, VCX, VCX-B, VCX-C, VCY, VDAC1, VEST1, Vgl-4, Vitrin, VPS13C, VWA5A, WDFY3, WDHD1, WDR18, WDR21A, WDR57, Wnt-8b, WWC2, XPF, XRCC1, XRCC2, XRCC4, XRCC6BP1, YFP, ZBTB24, ZBTB8B, ZC3H12B, ZCCHC4, ZCCHC7, ZFF29, ZFP108, ZFP109, ZFP11, ZFP110, ZFP113, ZFP119, ZFP141, ZFP192, ZFP213, ZFP26, ZFP37, Zfp870, ZNF100, ZNF33A, ZNF33B, ZNF510, ZNF596, ZNF746, ZNF772, ZNF818, ZRANB3, ZSCAN4.의 활성화자:
4930547C10Rik, 53BP1, A1-d, AIF, Aladin, ALKBH2, Apaf-1, APLF, APRIN, Arp9, ASF1A, ASF1B, ATR, Baxα, BC055324, BCLAF1, BinCARD, Bir1, BLM hydrolase, Bmf, BNIP-2, BNIPL-2, BRAF35, BTBD12, Bub2, C15orf41, C1orf227, C20orf196, C20orf43, C230052I12Rik, C330006K01Rik, CAD, caspase-3, caspase-8, CCDC6, CCDC76, Cdc13, Cdc25B, Cdc25C, Cdc2B, Cdc50C, CDCA2, Cdt2, CED-3, CED-6, CED-7, CED-8, CENP-S, Chk1, Chk2, Cid, Clb5, CPP32 p11, CPSF3, CPSF4, CRM1, Crt1, CTF8, CtIP, CTR9, cyclin δ-3, cyclin 2b, cyclin 3a, Cyclon, CYFIP2, D19Bwg1357e, DAP-3, DCC1, Dclre1c, DDB2, DDI1, DDI2, DDX1, DDX26B, DDX46, DEDD, DEDD2, DEM1, Diablo, Diva, DNA pol α, DNA pol ε A, DNase II, DR4, Dtl, EAF2, EG194588, EG240038, EG244495, EG333452, EG408191, EG434881, EGL-1, EI24, Elm1, Eme1, ENDOG, EndoV, ERCC1, Esp1, EST-1, EWS, EXOD1, FAM128A, FANCA, FANCD2, FANK1, FASP1, FHAD1, FIV gp36, FLASH, FLJ20433, Fnk, GADD 45β, GADD 45γ, GEN1, gp210, granzyme H, GTPBP8, H2bl2, Haspin, hCAP-G, Hfm1, Histone cluster 1 H2AE, Histone H2A.J, Histone H2A.X, hnRNP L, hnRNP X, HORMAD1, Hus1B, INO80C, Kar9, karyopherin α6, KCTD19, KIAA0406, Kin28, KRR1, Ku, La/SSB, LIN-35, LOC100041244, LOC641776, LOC643862, LOC643909, LOC729991, LRRC3B, LRWD1, MAGOHB, Mat1, MBD4, MCM5, MCM9, MCPyV large T-antigen, MDC1, MEI1, MLH3, MRE11_Mre11, MSH3_Msh3, MUS81, MYBBP1A, NAP1, Nek1, Nek8, NIRF, NSE1, NSE2, OR2G2, ORC2_Orc2, ORC3, OTTMUSG00000010552, OTTMUSG00000010657, OTUB1, p19 ARF, p53, p53CSV, p53R2, p73β, PA28α, PABP, PABPC2, PABPC6, PAR-4, PARP-1, PARP-8, Pdcd2L, PGBD3, PGBD5, PITSLRE A, PNK, POL H, Pol III RPC39, Pol2, POLA2, POLR2G, POLR2I, PQBP-1, Psf2, PSR, Rad23, Rad24, Rad26L, Rad3, Rad5, Rad51, Rad51B, Rad51D, Rad52, Rad57, Rad9, Rad9B, RAP80, Ras GAP, RBM4B, RecQL1, RecQL5, REV1, Rev3, REXO2, RFC, RFC1, RFC4, RFC5, RFWD3, RMI1, RMI2, RNase HII-A, RNase HII-C, RNase III Drosha, RNF113B, RNF168, RNF169, RP11-430L17.1, RP11-592B15.4, RPA 70 kDa subunit, RPRD1B, RREB1, RRP22, SFRS12, SFRS2B, SKIV2L, SLX1A/B_SLX1, Smac, SmarcAL1, SMC1α, SMC1β, SNM1B, SPACRCAN, SPATA22, SPATA5L1, SPHAR, SR140, SRp75, Srs2, SSBP4, String, Tβ-15b, TATDN2, TATDN3, TDRD3, TESSP2, TF, TFIIE-β, TFIIH p34, TGIF2LX1, Top1, Topo IIIβ, TP53INP1, TP53TG3, TP53TG5, TRIM46, Trp53i11, Trp53i13, TTC36, TTI1, TTP, U11/U12 snRNP 20K, UBE2T, UDG, USP51, VPRBP, Wee 1, Wip1, XPC, ZBTB37, ZNF347, ZNF440, ZNF8, ZW10.주문정보
| 제품명 | 카탈로그 번호 | 단위 | 가격 | 수량 | 관심품목 | |
Camptothecin, 50 mg | sc-200871 | 50 mg | $58.00 | |||
Camptothecin, 100 mg | sc-200871B | 100 mg | $94.00 | |||
Camptothecin, 250 mg | sc-200871A | 250 mg | $186.00 |
