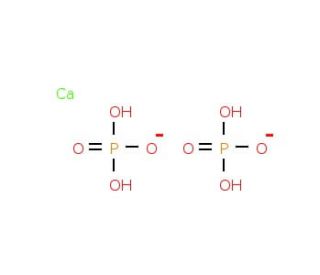
Calcium dihydrogenphosphate (CAS 7758-23-8)
QUICK LINKS
Calcium dihydrogenphosphate is an inorganic compound with far-reaching applications across various domains of science and industry. It′s employed extensively in creating fertilizers and food additives, and contributes to the manufacturing process of ceramics. The production of cement and concrete also involves the use of this compound. Calcium biphosphate is integral to several biological processes, such as bone mineralization and energy metabolism. In the realm of dentistry, it′s used for crafting dental composites, sealants, and other materials. Furthermore, it′s a vital element in the production of animal feed and pet food. In biological context, it′s a part of bone mineralization, contributing to the formation of hydroxyapatite, a key mineral component of bone. It′s involved in energy metabolism, providing a source of energy for cells. Moreover, it plays a role in pH regulation, being a source of bicarbonate ions that help maintain optimal pH levels.
Calcium dihydrogenphosphate (CAS 7758-23-8) References
- Effect of blending calcium compounds on hydrolytic degradation of poly(DL-lactic acid-co-glycolic acid). | Ara, M., et al. 2002. Biomaterials. 23: 2479-83. PMID: 12033595
- Evaluating the applicability of regulatory leaching tests for assessing the hazards of Pb-contaminated soils. | Halim, CE., et al. 2005. J Hazard Mater. 120: 101-11. PMID: 15811670
- The effect of tri-calcium phosphate (TCP) addition on the degradation of polylactide-co-glycolide (PLGA). | Ehrenfried, LM., et al. 2008. J Mater Sci Mater Med. 19: 459-66. PMID: 17607516
- Polymer assisted hydroxyapatite microspheres suitable for biomedical application. | Sinha, A., et al. 2008. J Mater Sci Mater Med. 19: 2009-13. PMID: 17952563
- Complete porphyrin analysis of pathological urines. | SVEINSSON, SL., et al. 1949. Scand J Clin Lab Invest. 1: 2-11. PMID: 18137001
- Hazardous animal waste carcasses transformation into slow release fertilizers. | Sharrock, P., et al. 2009. J Hazard Mater. 167: 119-23. PMID: 19185419
- Silver phosphate/graphitic carbon nitride as an efficient photocatalytic tandem system for oxygen evolution. | Yang, X., et al. 2015. ChemSusChem. 8: 1350-8. PMID: 25693743
- Phosphorus and nitrogen co-limitation of forest ground vegetation under elevated anthropogenic nitrogen deposition. | Hedwall, PO., et al. 2017. Oecologia. 185: 317-326. PMID: 28884383
- Impairing L-Threonine Catabolism Promotes Healthspan through Methylglyoxal-Mediated Proteohormesis. | Ravichandran, M., et al. 2018. Cell Metab. 27: 914-925.e5. PMID: 29551589
- [Determination of Ca/P atomic ratio in calcium-hydroxyapatite by neutron activation analysis]. | Mizumoto, Y. and Iwata, S. 1983. Radioisotopes. 32: 22-5. PMID: 6856880
- Effect of blending calcium compounds on hydrolytic degradation of poly (DL-lactic acid-co-glycolic acid) | M Ara, M Watanabe, Y Imai. 2002. Biomaterials. 23: 2479-2483.
- Development of radiophotoluminescence glass dosimeter usable in high temperature environment | F Sato, N Zushi, T Nagai, T Tanaka, Y Kato. 2013. Radiation Measurements. 53-54: 8-11.
- Interaction of calcium phosphates with calcium oxide or calcium hydroxide during the "soft" mechanochemical synthesis of hydroxyapatite | MV Chaikina, NV Bulina, OB Vinokurova. 2019. Ceramics International. 45: 16927-16933.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Calcium dihydrogenphosphate, 100 g | sc-252539 | 100 g | $88.00 | |||
Calcium dihydrogenphosphate, 500 g | sc-252539A | 500 g | $218.00 |
