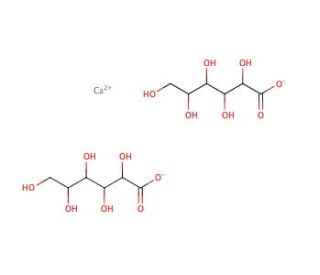
Calcium D-gluconate (CAS 299-28-5)
QUICK LINKS
Calcium D-gluconate is a compound utilized in the field of food science and nutrition research for its role as a calcium fortification agent. Studies involving this compound focus on optimizing the bioavailability of calcium in various food matrices, enhancing the nutritional value of processed foods. In the area of materials research, Calcium D-gluconate is explored for its potential in the formulation of biodegradable packaging materials that could release calcium ions, which have antimicrobial properties. Additionally, it is used in agricultural research to investigate its effects as a plant nutrient supplement, potentially influencing plant growth and crop yield. Calcium D-gluconate is also examined for its ability to act as a chelating agent in the removal of heavy metals from contaminated soils or wastewater through the formation of stable calcium complexes.
Calcium D-gluconate (CAS 299-28-5) References
- Determination of L-threonate in calcium L-threonate preparations by capillary electrophoresis with indirect UV detection. | Song, JZ., et al. 1999. Electrophoresis. 20: 1850-5. PMID: 10445326
- [On the spectrocolorimetric determination of calcium-D-gluconate]. | Illner, E. 1975. Pharmazie. 30: 407. PMID: 1161797
- D-Gluconic Acid: Isolation from the Defensive Secretion of the Cockroach Eurycotis decipiens. | Dateo, GP. and Roth, LM. 1967. Science. 155: 88-9. PMID: 17799153
- Synthesis of sintering-resistant sorbents for CO2 capture. | Liu, W., et al. 2010. Environ Sci Technol. 44: 3093-7. PMID: 20205453
- Aqueous solubility of calcium L-lactate, calcium D-gluconate, and calcium D-lactobionate: importance of complex formation for solubility increase by hydroxycarboxylate mixtures. | Vavrusova, M., et al. 2013. J Agric Food Chem. 61: 8207-14. PMID: 23906043
- Spontaneous supersaturation of calcium D-gluconate during isothermal dissolution of calcium L-lactate in aqueous sodium D-gluconate. | Vavrusova, M. and Skibsted, LH. 2014. Food Funct. 5: 85-91. PMID: 24281051
- Thermodynamics of dissolution of calcium hydroxycarboxylates in water. | Vavrusova, M., et al. 2014. J Agric Food Chem. 62: 5675-81. PMID: 24869479
- Calcium d-Saccharate: Aqueous Solubility, Complex Formation, and Stabilization of Supersaturation. | Garcia, AC., et al. 2016. J Agric Food Chem. 64: 2352-60. PMID: 26934422
- Block-copolymer-assisted synthesis of hydroxyapatite nanoparticles with high surface area and uniform size. | Huang, YT., et al. 2011. Sci Technol Adv Mater. 12: 045005. PMID: 27877424
- Slow lactate gluconate exchange in calcium complexes during precipitation from supersaturated aqueous solutions. | Garcia, AC., et al. 2020. Food Res Int. 137: 109539. PMID: 33233167
- Fabrication and Use of Dry Macroporous Alginate Scaffolds for Viral Transduction of T Cells. | VanBlunk, M., et al. 2022. J Vis Exp.. PMID: 36156536
- Effects of various parameters on solution-mediated phase transformation of calcium d-gluconate: an approach to obtain pure metastable monohydrate. | Zhang, H., et al. 2023. RSC Adv. 13: 12175-12183. PMID: 37091620
- [Determination of d-glucose in calcium-d-gluconate and the analysis of calcium-d-gluconate]. | Laass, W. 1973. Pharmazie. 28: 684. PMID: 4783561
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Calcium D-gluconate, 100 g | sc-221393 | 100 g | $40.00 | |||
Calcium D-gluconate, 500 g | sc-221393A | 500 g | $145.00 |
