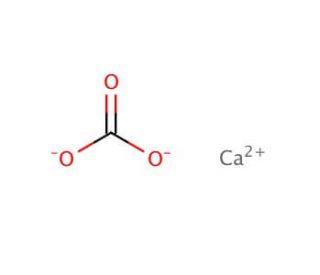
Calcium carbonate (CAS 471-34-1)
See product citations (1)
QUICK LINKS
Calcium carbonate (CaCO3) is a naturally occurring mineral and one of the most abundant substances on earth, found in rocks such as limestone and chalk, and in biological materials such as shellfish shells and coral. It plays a pivotal role in various scientific fields, notably in geochemistry, environmental science, and materials science. In geochemical research, calcium carbonate is crucial for studying biogeochemical cycles, particularly the carbon cycle, where it acts as a major carbon sink. The precipitation and dissolution of calcium carbonate in oceanic environments are vital processes that regulate atmospheric CO2 levels and oceanic acidity. In environmental studies, calcium carbonate is used to mitigate acidity in soils and water bodies, a practice essential for restoring ecological balance and promoting healthy aquatic life. Additionally, its use in carbon sequestration projects highlights its potential in combating climate change by capturing carbon dioxide. In materials science, calcium carbonate serves as a primary component in the manufacture of cement and as a filler in the plastics, paper, and paint industries, where it improves the properties of materials, such as brightness and surface smoothness. Furthermore, its role in the production of glass and ceramic products showcases its versatility and importance in industrial applications. Through these diverse uses, calcium carbonate is instrumental in advancing research and industrial practices, contributing significantly to environmental management and material enhancement.
Calcium carbonate (CAS 471-34-1) References
- Comparison of calcium acetate with calcium carbonate as phosphate binder in patients on maintenance haemodialysis. | Saif, I., et al. 2007. J Ayub Med Coll Abbottabad. 19: 26-8. PMID: 18693591
- Raloxifene plus ossein-hydroxyapatite compound versus raloxifene plus calcium carbonate to control bone loss in postmenopausal women: a randomized trial. | Pelayo, I., et al. 2008. Menopause. 15: 1132-8. PMID: 18791486
- Application of microbially induced calcium carbonate precipitation in designing bio self-healing concrete. | Seifan, M. and Berenjian, A. 2018. World J Microbiol Biotechnol. 34: 168. PMID: 30387067
- Calcium carbonate: Adored and ignored in bioactivity assessment. | Mozafari, M., et al. 2019. Acta Biomater. 91: 35-47. PMID: 31004843
- Microbially induced calcium carbonate precipitation: a widespread phenomenon in the biological world. | Seifan, M. and Berenjian, A. 2019. Appl Microbiol Biotechnol. 103: 4693-4708. PMID: 31076835
- Calcium Carbonate Formation in the Presence of Biopolymeric Additives. | Azulay, DN. and Chai, L. 2019. J Vis Exp.. PMID: 31157787
- Calcium carbonate supplementation to chorioallantoic membranes improves hatchability in shell-less chick embryo culture. | Tahara, Y., et al. 2021. J Biosci Bioeng. 131: 314-319. PMID: 33223431
- Calcium carbonate supplementation causes motor dysfunction. | Sugiura, A., et al. 2022. Exp Anim. 71: 399-410. PMID: 35584940
- Calcium carbonate occluding a peritoneal dialysis catheter. | Mitsuno, R., et al. 2023. Clin Exp Nephrol. 27: 96-97. PMID: 36088526
- Microbially-Induced Calcium Carbonate Precipitation Test on Yellow Sandstone Based on LF-NMR Monitoring. | Zhuang, C., et al. 2022. Int J Environ Res Public Health. 19: PMID: 36554738
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Calcium carbonate, 100 g | sc-203325 | 100 g | $31.00 | |||
Calcium carbonate, 500 g | sc-203325A | 500 g | $102.00 |
