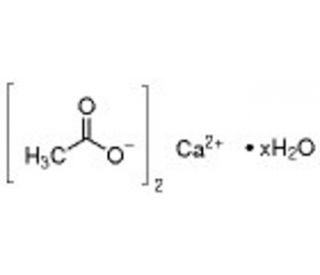
Molecular structure of Calcium acetate hydrate, CAS Number: 114460-21-8
Calcium acetate hydrate (CAS 114460-21-8)
CAS Number:
114460-21-8
Purity:
99%
Molecular Weight:
158.17 (anhydrous basis)
Molecular Formula:
C4H8CaO5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Calcium acetate hydrate, also referred to as calcium ethanoate, is an organic compound comprising calcium and acetic acid. It exists as a white, odorless, crystalline powder with water solubility and insolubility in alcohol. Calcium acetate hydrate serves as a buffering agent in biochemical and physiological experiments, a reagent in organic synthesis, and a chelating agent. Additionally, it contributes to the production of calcium acetate carbonate, which finds use in the manufacturing of cement, paper, and various other products.
Calcium acetate hydrate (CAS 114460-21-8) References
- A spectroscopic study of the mineral paceite (calcium acetate). | Musumeci, AW., et al. 2007. Spectrochim Acta A Mol Biomol Spectrosc. 67: 649-61. PMID: 17070100
- Calcium precursors for the production of CaO sorbents for multicycle CO2 capture. | Liu, W., et al. 2010. Environ Sci Technol. 44: 841-7. PMID: 20030311
- Synthesis of sintering-resistant sorbents for CO2 capture. | Liu, W., et al. 2010. Environ Sci Technol. 44: 3093-7. PMID: 20205453
- Corrosion of Heritage Objects: Collagen-Like Triple Helix Found in the Calcium Acetate Hemihydrate Crystal Structure. | Bette, S., et al. 2020. Angew Chem Int Ed Engl. 59: 9438-9442. PMID: 32065724
- Synthesis, spectroscopic properties, and x-ray structure of [1, 7-bis (5-methylimidazol-4-yl)-2, 6-dithiaheptane] dichlorocopper (II): a compound with unusually long copper (II)-thioether bonds | Van Rijn, J., Driessen, W. L., Reedijk, J., & Lehn, J. M. 1984. Inorganic Chemistry. 23(22): 3584-3588.
- The effect of fluoride treatment on titanium treated with anodic spark oxidation | Park, I. S., Kim, J. J., Ahn, S. G., Lee, M. H., Seol, K. W., & Bae, T. S. 2007. Metals and Materials International. 13: 117-122.
- Dielectric and ferroelectric studies on sol-gel derived calcium modified lead zirconate titanate ceramics | Sachdeva, A., Luthra, V., Gautam, P., & Tandon, R. P. 2010. Integrated Ferroelectrics. 122(1): 74-82.
- Pharmacokinetic characterization of calcium from three calcium salts (calcium chloride, calcium acetate and calcium ascorbate) in mice | Ueda, Y., & Taira, Z. 2012. Journal of Hard Tissue Biology. 21(3): 291-298.
- Characteristics of multi-layer coatings synthesized on Ti6Al4V alloy by micro-arc oxidation in silver nitrate added electrolytes | Muhaffel, F., Cempura, G., Menekse, M., Czyrska-Filemonowicz, A., Karaguler, N., & Cimenoglu, H. 2016. Surface and Coatings Technology. 307: 308-315.
- Surface modification of severe plastically deformed ultrafine grained pure titanium by plasma electrolytic oxidation | Reshadi, F., Faraji, G., Baniassadi, M., & Tajeddini, M. 2017. Surface and Coatings Technology. 316: 113-121.
- Calcium phosphate powder synthesized from calcium acetate and ammonium hydrophosphate for bioceramics application | Safronova, T., Putlayev, V., Filippov, Y., Shatalova, T., Karpushkin, E., Larionov, D.,.. & Shakhtarin, Y. 2018. Ceramics. 1(2): 375-392.
- Structural Studies of Orotate Phosphoribosyl Transferase (TTHA1742) from Hyperthermophilic Thermus thermophilus HB8 | Mutharasappan, N., & Jeyaraman, J. 2019. SPring-8/SACLA Research Report. 7(2): 125-129.
- In situ upgrading of bio-oil via CaO catalyst derived from organic precursors | Yi, L., Liu, H., Xiao, K., Wang, G., Zhang, Q., Hu, H., & Yao, H. 2019. Proceedings of the Combustion Institute. 37(3): 3119-3126.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Calcium acetate hydrate, 100 g | sc-255008 | 100 g | $47.00 | |||
Calcium acetate hydrate, 500 g | sc-255008A | 500 g | $62.00 | |||
Calcium acetate hydrate, 1 kg | sc-255008B | 1 kg | $109.00 |
