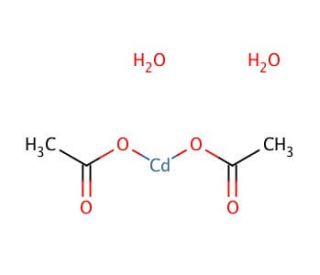
Molecular structure of Cadmium acetate dihydrate, CAS Number: 5743-04-4
Cadmium acetate dihydrate (CAS 5743-04-4)
CAS Number:
5743-04-4
Purity:
98%
Molecular Weight:
266.53
Molecular Formula:
C4H10CdO6
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Cadmium acetate dihydrate is an inorganic salt that exhibits solubility in water, ethanol, and methanol. It finds use in scientific research, particularly in the biochemistry field, serving as a reagent for the synthesis of diverse compounds. Additionally, it acts as a catalyst in organic synthesis. Its applications extend to the synthesis of cadmium-containing polymers, dyes, and organometallic compounds.
Cadmium acetate dihydrate (CAS 5743-04-4) References
- Revisiting the calculation of (13)C chemical shift tensors in cadmium acetate dihydrate with EIM and EIM/cluster methods. | Orendt, AM. 2006. Magn Reson Chem. 44: 385-9. PMID: 16477671
- Use of ionic liquids in the synthesis of nanocrystals and nanorods of semiconducting metal chalcogenides. | Biswas, K. and Rao, CN. 2007. Chemistry. 13: 6123-9. PMID: 17497619
- CdS magic-sized nanocrystals exhibiting bright band gap photoemission via thermodynamically driven formation. | Li, M., et al. 2009. ACS Nano. 3: 3832-8. PMID: 19911809
- Bis{4-phenyl-1-[1-(pyridin-2-yl-κN)ethyl-idene]thio-semicarbazidato-κ(2)N(1),S}cadmium. | Fonseca, Ade S., et al. 2012. Acta Crystallogr Sect E Struct Rep Online. 68: m635-6. PMID: 22590129
- Photoluminescent enhancement of CdSe/Cd(1-x) Zn(x)S quantum dots by hexadecylamine at room temperature. | Yang, J. and Yang, P. 2012. J Nanosci Nanotechnol. 12: 7322-8. PMID: 23035471
- Quantum dots sensitized titanium dioxide decorated reduced graphene oxide for visible light excited photoelectrochemical biosensing at a low potential. | Zeng, X., et al. 2014. Biosens Bioelectron. 54: 331-8. PMID: 24291752
- Fabrication and characterization of ZnO@CdS core-shell nanostructure using acetate precursors: XRD, FESEM, DRS, FTIR studies and effects of cadmium ion concentration on band gap. | Habibi, MH. and Rahmati, MH. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 133: 13-8. PMID: 24926644
- Crystal structure of cis-bis-{4-phenyl-1-[(3R)-1,7,7-tri-methyl-2-oxobi-cyclo-[2.2.1]heptan-3-ylidene]thio-semicarbazidato-κ(3) O,N (1),S}cadmium(II) with an unknown solvent mol-ecule. | Nogueira, VS., et al. 2015. Acta Crystallogr E Crystallogr Commun. 71: m234-5. PMID: 26870441
- Transparent conducting ZnO-CdO mixed oxide thin films grown by the sol-gel method. | Pathak, TK., et al. 2017. J Colloid Interface Sci. 487: 378-387. PMID: 27810506
- Comparative assessment of the effects of meso-2,3-dimercaptosuccinic acid and salinomycin on spleen function of cadmium-exposed mice. | Kamenova, K., et al. 2019. Environ Sci Pollut Res Int. 26: 33304-33310. PMID: 31520384
- Hierarchically Porous WO3/CdWO4 Fiber-in-Tube Nanostructures Featuring Readily Accessible Active Sites and Enhanced Photocatalytic Effectiveness for Antibiotic Degradation in Water. | Rong, F., et al. 2021. ACS Appl Mater Interfaces. 13: 21138-21148. PMID: 33908249
- Hollow CdS-ZnS-ZIF-8 Polyhedron for Visible Light-Induced Cr(VI) Reduction. | Wang, J., et al. 2023. Inorg Chem. 62: 1047-1053. PMID: 36580399
- Performance improvement of dye sensitized solar cells based on cadmium sulfide/S, N co doped carbon dots nanocomposites | H Safardoust-Hojaghan, O Amiri. 2020. Journal of Molecular Liquids. 301: 112413.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cadmium acetate dihydrate, 100 g | sc-210996 | 100 g | $60.00 | |||
Cadmium acetate dihydrate, 500 g | sc-210996A | 500 g | $227.00 |
