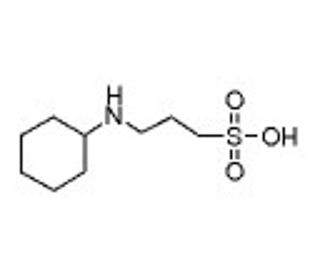
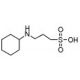
CABS (CAS 161308-34-5)
See product citations (4)
QUICK LINKS
CABS, chemically known as ChloroAmidoBenzeneSulfonamide, is an intriguing compound utilized predominantly in the field of organic chemistry research. This molecule, characterized by a benzene ring substituted with a sulfonamide group and a chloro substituent, serves as a critical reagent in the synthesis of more complex chemical entities. The sulfonamide group plays a pivotal role, often involved in the creation of hydrogen bonds due to its ability to act as both a hydrogen bond donor and acceptor. This dual capability enhances its utility in modulating the solubility and stability of potential products. The chloro group, on the other hand, can undergo various chemical reactions, such as nucleophilic substitution, making CABS a versatile starting material for introducing other functional groups during synthetic processes. Researchers have employed CABS in the synthesis of diverse sulfonamide derivatives, exploring its applications in creating new materials with potential industrial applications. For example, it has been used in the development of novel polymers and coatings where enhanced durability and chemical resistance are desired. In addition to its synthetic utility, the structural features of CABS allow it to be studied in computational chemistry for understanding molecular interactions and reaction mechanisms, further broadening its application spectrum in scientific research.
CABS (CAS 161308-34-5) References
- Validation and long-term assessment of an approach for the high throughput determination of lipophilicity (log POW) values using multiplexed, absorbance-based capillary electrophoresis. | Wong, KS., et al. 2004. J Pharm Sci. 93: 916-31. PMID: 14999729
- Novel role of tyrosine in catalysis by human AP endonuclease 1. | Mundle, ST., et al. 2004. DNA Repair (Amst). 3: 1447-55. PMID: 15380100
- The zinspy family of fluorescent zinc sensors: syntheses and spectroscopic investigations. | Nolan, EM. and Lippard, SJ. 2004. Inorg Chem. 43: 8310-7. PMID: 15606177
- Selective Hg(II) detection in aqueous solution with thiol derivatized fluoresceins. | Nolan, EM., et al. 2006. Inorg Chem. 45: 2742-9. PMID: 16529499
- Turn-on and ratiometric mercury sensing in water with a red-emitting probe. | Nolan, EM. and Lippard, SJ. 2007. J Am Chem Soc. 129: 5910-8. PMID: 17429971
- The foldon substructure of staphylococcal nuclease. | Bédard, S., et al. 2008. J Mol Biol. 376: 1142-54. PMID: 18201720
- Evaluating the potential for enzymatic acrylamide mitigation in a range of food products using an asparaginase from Aspergillus oryzae. | Hendriksen, HV., et al. 2009. J Agric Food Chem. 57: 4168-76. PMID: 19388639
- The Trypanosoma cruzi virulence factor oligopeptidase B (OPBTc) assembles into an active and stable dimer. | Motta, FN., et al. 2012. PLoS One. 7: e30431. PMID: 22276197
- (Un) suitability of the use of pH buffers in biological, biochemical and environmental studies and their interaction with metal ions–a review[J]. | Ferreira C M H, Pinto I S S, Soares E V. 2015,. Rsc Advances,. 5(39):: 30989-31003.
- Synthesis and properties of water-dispersible polyisocyanates carrying sulfonate[J]. | Peng Z, Tang L, Yuan Y,. 2020. Journal of Coatings Technology and Research,., 17:: 345-359.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
CABS, 25 g | sc-285279 | 25 g | $460.00 |
