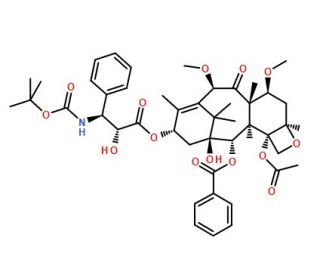
Molecular structure of Cabazitaxel, CAS Number: 183133-96-2
Cabazitaxel (CAS 183133-96-2)
See product citations (1)
Alternate Names:
Jevtana; XRP-6258
Application:
Cabazitaxel is a semi-synthetic toxal derivative
CAS Number:
183133-96-2
Purity:
≥98%
Molecular Weight:
835.93
Molecular Formula:
C45H57NO14
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Cabazitaxel is a semi-synthetic derivative of toxal (sc-201439), a natural taxoid microtubule inhibitor. Cabazitaxel may act to have antitumor activity in vitro, and while it has a lower affinity for the ATP-dependent efflux pump, P-glycoprotein 1 (P-gp) relative to toxal (sc-201439) or docetaxel (sc-201436A), it has shown greater ability to penetrate the blood-brain barrier than either. In mice and rat models, uptake of cabazitaxel may act to increase at concentrations above 11 muM and saturation has been seen at 13 muM. This seems to suggest that P-gp may be essential to transport cabazitaxel across the blood-brain barrier at specific threshold concentrations.
Cabazitaxel (CAS 183133-96-2) References
- Cabazitaxel. | Galsky, MD., et al. 2010. Nat Rev Drug Discov. 9: 677-8. PMID: 20811375
- Cabazitaxel, a new taxane with favorable properties. | Bouchet, BP. and Galmarini, CM. 2010. Drugs Today (Barc). 46: 735-42. PMID: 21076710
- Cabazitaxel for the treatment of prostate cancer. | Michielsen, DP., et al. 2011. Expert Opin Pharmacother. 12: 977-82. PMID: 21406025
- Cabazitaxel: a novel second-line treatment for metastatic castration-resistant prostate cancer. | Paller, CJ. and Antonarakis, ES. 2011. Drug Des Devel Ther. 5: 117-24. PMID: 21448449
- Cabazitaxel: a novel microtubule inhibitor. | Villanueva, C., et al. 2011. Drugs. 71: 1251-8. PMID: 21770474
- Cabazitaxel in metastatic castration-resistant prostate cancer. | Yap, TA., et al. 2012. Expert Rev Anticancer Ther. 12: 1129-36. PMID: 23098113
- Preclinical profile of cabazitaxel. | Vrignaud, P., et al. 2014. Drug Des Devel Ther. 8: 1851-67. PMID: 25378905
- Clinical concepts for cabazitaxel in the management of metastatic castration-resistant prostate cancer. | Al-Mansouri, L. and Gurney, H. 2019. Asia Pac J Clin Oncol. 15: 288-295. PMID: 31313526
- Cabazitaxel plus carboplatin for the treatment of men with metastatic castration-resistant prostate cancers: a randomised, open-label, phase 1-2 trial. | Corn, PG., et al. 2019. Lancet Oncol. 20: 1432-1443. PMID: 31515154
- Cabazitaxel schedules in metastatic castration-resistant prostate cancer: a review. | Pobel, C., et al. 2021. Future Oncol. 17: 91-102. PMID: 33463373
- [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. | Hofman, MS., et al. 2021. Lancet. 397: 797-804. PMID: 33581798
- Effect of cabazitaxel on macrophages improves CD47-targeted immunotherapy for triple-negative breast cancer. | Cao, X., et al. 2021. J Immunother Cancer. 9: PMID: 33753567
- Real-World Cabazitaxel Use and Outcomes in Metastatic Castrate-Resistant Prostate Cancer: The Impact of Response to First ARPI. | Watson, AS., et al. 2022. Clin Genitourin Cancer. 20: 496.e1-496.e9. PMID: 35599196
- Current development of cabazitaxel drug delivery systems. | Sun, B., et al. 2023. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 15: e1854. PMID: 36161272
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cabazitaxel, 5 mg | sc-396754 | 5 mg | $79.00 |
