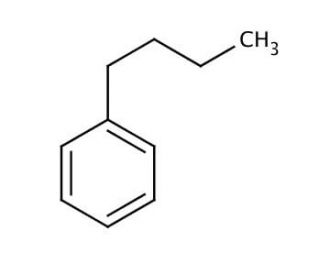
Butylbenzene (CAS 104-51-8)
QUICK LINKS
Butylbenzene is an organic chemical compound characterized by a linear four-carbon butyl group attached to a benzene ring. This structure places it within the alkylbenzene family, where the hydrophobic alkyl chain imparts lipophilic properties while the aromatic ring contributes to its ability to engage in π-π interactions, making it a non-polar solvent with a moderate boiling point. In research, butylbenzene is primarily used as a solvent or chemical intermediate due to its ability to dissolve non-polar substances and participate in organic synthesis. Its mechanism of action as a solvent stems from van der Waals forces, which facilitate the dissolution of other non-polar compounds, making it useful in a variety of chemical processes, including the purification of non-polar materials and in the extraction of organic compounds. Butylbenzene′s relatively inert nature also makes it a choice solvent in the study of reaction mechanisms, especially where the solvent′s non-reactivity is crucial for unbiased observations. In polymer science, it is often employed in the synthesis and study of polymers, where its solubility parameters are matched with those of the polymer to effectively dissolve and manipulate polymer chains. Additionally, the compound′s structure has made it a subject in the investigation of hydrophobic interactions and the thermodynamics of solute-solvent systems, providing insights into the behavior of similar alkyl-substituted aromatic compounds in various industrial and research applications.
Butylbenzene (CAS 104-51-8) References
- Internal energy content of n-butylbenzene, bromobenzene, iodobenzene and aniline molecular ions generated by two-photon ionization at 266 nm. A photodissociation study. | Yoon, OK., et al. 1999. Rapid Commun Mass Spectrom. 13: 1515-21. PMID: 10407348
- Uterotrophic and Hershberger assays for n-butylbenzene in rats. | Yamasaki, K., et al. 2002. Arch Toxicol. 75: 703-6. PMID: 11876503
- Translational to vibrational energy conversion during surface-induced dissociation of n-butylbenzene molecular ions colliding at self-assembled monolayer surfaces. | Jo, SC. and Cooks, RG. 2003. Eur J Mass Spectrom (Chichester). 9: 237-4. PMID: 12939476
- Organic solvent-induced cell death in rat cerebellar granule cells: structure dependence of c10 hydrocarbons and relationship to reactive oxygen species formation. | Dreiem, A., et al. 2005. Neurotoxicology. 26: 321-30. PMID: 15935204
- A two-generation reproductive toxicity study of n-butylbenzene in rats. | Izumi, H., et al. 2005. J Toxicol Sci. 30 Spec No.: 21-38. PMID: 16641541
- Comparison of two vectors for functional expression of a bacterial cytochrome P450 gene in Escherichia coli using CYP153 genes. | Fujita, N., et al. 2009. Biosci Biotechnol Biochem. 73: 1825-30. PMID: 19661686
- Anharmonic modeling of the conformation-specific IR spectra of ethyl, n-propyl, and n-butylbenzene. | Tabor, DP., et al. 2016. J Chem Phys. 144: 224310. PMID: 27306010
- Butylbenzene and tert-Butylbenzene-Sorption on Sand Particles and Biodegradation in the Presence of Plant Natural Surfactants. | Zdarta, A., et al. 2018. Toxins (Basel). 10: PMID: 30131465
- Bioactivities of 3-Butylidenephthalide and n-Butylbenzene from the Essential Oil ofLigusticum jeholense against Stored-product Insects. | Luo, C., et al. 2019. J Oleo Sci. 68: 931-937. PMID: 31413242
- Orderly MOF-Assembled Hybrid Monolithic Stationary Phases for Nano-Flow HPLC. | Ding, M., et al. 2020. Anal Chem. 92: 15757-15765. PMID: 33253547
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Butylbenzene, 5 ml | sc-234246 | 5 ml | $42.00 |
