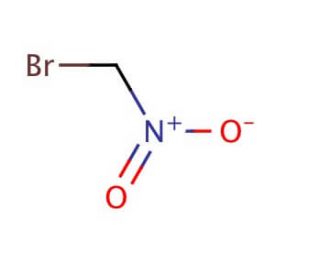
Bromonitromethane (CAS 563-70-2)
QUICK LINKS
Bromonitromethane is a chemical compound that functions as an alkylating agent in development settings. Its mode of action involves the alkylation of nucleophilic sites on biomolecules, such as DNA and proteins, leading to the formation of covalent bonds. Can modify the structure and function of biomolecules, potentially impacting cellular processes and signaling pathways. Bromonitromethane′s reactivity with biological molecules makes it a useful for studying the effects of alkylation on cellular function and for investigating the mechanisms of chemical-induced DNA damage. Its ability to covalently modify biomolecules provides insights into the potential biological consequences of exposure to alkylating agents and aids in understanding the molecular basis of chemical toxicity. In experimental, bromonitromethane′s mode of action as an alkylating agent contributes to the study of chemical-induced biological effects and the development of strategies for mitigating chemical exposure.
Bromonitromethane (CAS 563-70-2) References
- Efficient addition reaction of bromonitromethane to aldehydes catalyzed by NaI: a new route to 1-bromo-1-nitroalkan-2-ols under very mild conditions. | Concellón, JM., et al. 2006. Org Lett. 8: 5979-82. PMID: 17165909
- Stereoselective NaN3-catalyzed halonitroaldol-type reaction of azetidine-2,3-diones in aqueous media. | Alcaide, B., et al. 2008. Org Biomol Chem. 6: 1635-40. PMID: 18421397
- Genotoxicity analysis of two halonitromethanes, a novel group of disinfection by-products (DBPs), in human cells treated in vitro. | Liviac, D., et al. 2009. Environ Res. 109: 232-8. PMID: 19200951
- Free-radical chemistry of disinfection byproducts. 3. Degradation mechanisms of chloronitromethane, bromonitromethane, and dichloronitromethane. | Mincher, BJ., et al. 2010. J Phys Chem A. 114: 117-25. PMID: 20055512
- Toxicity profile of labile preservative bronopol in water: the role of more persistent and toxic transformation products. | Cui, N., et al. 2011. Environ Pollut. 159: 609-15. PMID: 21035931
- Evidence for chemical and cellular reactivities of the formaldehyde releaser bronopol, independent of formaldehyde release. | Kireche, M., et al. 2011. Chem Res Toxicol. 24: 2115-28. PMID: 22034943
- Stereoselective synthesis of highly functionalized nitrocyclopropanes through the organocatalyic Michael-addition-initiated cyclization of bromonitromethane and β,γ-unsaturated α-ketoesters. | Yu, H., et al. 2013. Chem Asian J. 8: 2859-63. PMID: 23934622
- Synthesis of enantiopure 2-C-glycosyl-3-nitrochromenes. | Soengas, RG., et al. 2013. J Org Chem. 78: 12831-6. PMID: 24279432
- Enantioselective Synthesis of D-α-Amino Amides from Aliphatic Aldehydes. | Schwieter, KE. and Johnston, JN. 2015. Chem Sci. 6: 2590-2595. PMID: 25838883
- Enantioselective Addition of Bromonitromethane to Aliphatic N-Boc Aldimines Using a Homogeneous Bifunctional Chiral Organocatalyst. | Schwieter, KE. and Johnston, JN. 2015. ACS Catal. 5: 6559-6562. PMID: 27019764
- In vitro studies on the tumorigenic potential of the halonitromethanes trichloronitromethane and bromonitromethane. | Marsà, A., et al. 2017. Toxicol In Vitro. 45: 72-80. PMID: 28844819
- Rapid and complete dehalogenation of halonitromethanes in simulated gastrointestinal tract and its influence on toxicity. | Yin, J., et al. 2018. Chemosphere. 211: 1147-1155. PMID: 30223330
- Resolving Bromonitromethane Sourcing by Synthesis: Preparation at the Decagram Scale. | Thorpe, MP., et al. 2022. J Org Chem. 87: 5451-5455. PMID: 35364809
- Structural and functional alterations of intestinal flora in mice induced by halonitromethanes exposure. | Yin, J., et al. 2022. Front Microbiol. 13: 991818. PMID: 36177464
- Thio-sugars. IV: Design and synthesis of S-linked fucoside analogs as a new class of alpha-L-fucosidase inhibitors. | Witczak, ZJ. and Boryczewski, D. 1998. Bioorg Med Chem Lett. 8: 3265-8. PMID: 9873715
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromonitromethane, 1 g | sc-234219 | 1 g | $37.00 | |||
Bromonitromethane, 10 g | sc-234219A | 10 g | $94.00 |
