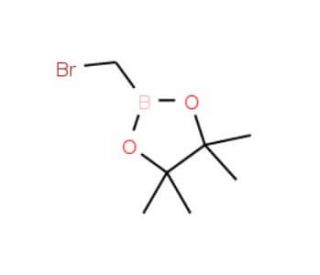
Bromomethylboronic acid pinacol ester (CAS 166330-03-6)
QUICK LINKS
Bromomethylboronic acid pinacol ester or also known as 2-(Bromomethyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (BMTMD) is an organoboron compound extensively researched for its potential applications in organic synthesis and catalysis. It serves as a versatile reagent, exhibiting a broad range of reactivity suitable for various reactions, including Diels-Alder, Michael, and Wittig reactions. Moreover, Bromomethylboronic acid pinacol ester proves valuable as a catalyst in the synthesis of diverse compounds like heterocycles, amines, polymers (such as polyesters and polyamides), and organometallic compounds (including boronates, boronic esters, and boronic acids). Its ability to react with various functional groups, including carbonyl and carboxyl groups, facilitates nucleophilic addition and substitution mechanisms, respectively. Additionally, Bromomethylboronic acid pinacol ester can interact with amines, alcohols, and sulfonates, where nucleophilic addition and substitution mechanisms are involved depending on the functional group.
Bromomethylboronic acid pinacol ester (CAS 166330-03-6) References
- Stereocontrolled synthesis of adjacent acyclic quaternary-tertiary motifs: application to a concise total synthesis of (-)-filiformin. | Blair, DJ., et al. 2014. Angew Chem Int Ed Engl. 53: 5552-5. PMID: 24757079
- A near-infrared probe for non-invasively monitoring cerebrospinal fluid flow by 18F-positron emitting tomography and fluorescence. | Guo, H., et al. 2020. EJNMMI Res. 10: 37. PMID: 32301036
- A Boronated Derivative of Temozolomide Showing Enhanced Efficacy in Boron Neutron Capture Therapy of Glioblastoma. | Xiang, J., et al. 2022. Cells. 11: PMID: 35406737
- The total synthesis of (−)-aplysin via a lithiation–borylation–propenylation sequence | Catherine J. Fletcher a, Daniel J. Blair a, Katherine M.P. Wheelhouse b, Varinder K. Aggarwal a. 7598-7604. Tetrahedron. 68: 2012.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromomethylboronic acid pinacol ester, 1 g | sc-263062 | 1 g | $51.00 | |||
Bromomethylboronic acid pinacol ester, 5 g | sc-263062A | 5 g | $163.00 |
