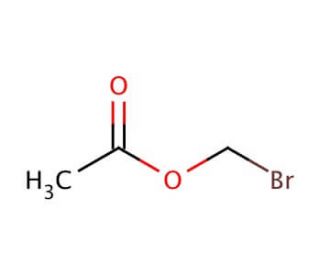
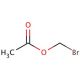
Molecular structure of Bromomethyl Acetate, CAS Number: 590-97-6
Bromomethyl Acetate (CAS 590-97-6)
CAS Number:
590-97-6
Molecular Weight:
152.97
Molecular Formula:
C3H5BrO2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Bromomethyl Acetate has cytotoxic and mutagenic effects. It is used in the synthesis of a variety of organic compounds, including alcohols, esters, and amines.. Additionally, bromomethyl acetate has been used in the synthesis of a variety of heterocyclic compounds, such as thiophenes, pyrroles, and furans. bromomethyl acetate mechanism is based on the formation of a bromomethyl anion from the reaction of bromoacetic acid and methanol. The bromomethyl anion then reacts with the desired substrate, forming a new compound.
Bromomethyl Acetate (CAS 590-97-6) References
- Esterase-sensitive nitric oxide donors of the diazeniumdiolate family: in vitro antileukemic activity. | Saavedra, JE., et al. 2000. J Med Chem. 43: 261-9. PMID: 10649981
- Synthesis and evaluation of iron chelators with masked hydrophilic moieties. | Meijler, MM., et al. 2002. J Am Chem Soc. 124: 12666-7. PMID: 12392406
- New Ca2+ fluoroionophores based on the BODIPY fluorophore. | Gee, KR., et al. 2003. Comb Chem High Throughput Screen. 6: 363-6. PMID: 12769680
- O6-alkylguanine-DNA alkyltransferase has opposing effects in modulating the genotoxicity of dibromomethane and bromomethyl acetate. | Liu, L., et al. 2004. Chem Res Toxicol. 17: 742-52. PMID: 15206895
- Optically active cyclohexene derivative as a new antisepsis agent: an efficient synthesis of ethyl (6R)-6-[N-(2-chloro-4-fluorophenyl)sulfamoyl]cyclohex-1-ene-1-carboxylate (TAK-242). | Yamada, M., et al. 2006. Chem Pharm Bull (Tokyo). 54: 58-62. PMID: 16394550
- The effect of structure on nitroxide EPR spectral linewidth. | Burks, SR., et al. 2010. J Org Chem. 75: 4737-41. PMID: 20540511
- Convergent synthesis of trisubstituted Z-allylic esters by Wittig-Schlosser reaction. | Hodgson, DM. and Arif, T. 2010. Org Lett. 12: 4204-7. PMID: 20722384
- Esterification of an unnatural amino acid structurally deviating from canonical amino acids promotes its uptake and incorporation into proteins in mammalian cells. | Takimoto, JK., et al. 2010. Chembiochem. 11: 2268-72. PMID: 20872394
- Lanthanide complex-based luminescent probes for highly sensitive time-gated luminescence detection of hypochlorous acid. | Xiao, Y., et al. 2012. Anal Chem. 84: 10785-92. PMID: 23190019
- Room-Temperature Electron Spin Relaxation of Triarylmethyl Radicals at the X- and Q-Bands. | Kuzhelev, AA., et al. 2015. J Phys Chem B. 119: 13630-13640. PMID: 26001103
- Automated access to well-defined ionic oligosaccharides. | Zhu, Y., et al. 2020. Org Biomol Chem. 18: 1349-1353. PMID: 32037424
- Caged nitric oxide. Stable organic molecules from which nitric oxide can be photoreleased. | Makings, LR. and Tsien, RY. 1994. J Biol Chem. 269: 6282-5. PMID: 8119976
- Metabolically convertible lipophilic derivatives of pH-sensitive amphipathic photosensitizers. | Sahai, D., et al. 1993. Photochem Photobiol. 58: 803-8. PMID: 8309999
- Expression of mammalian glutathione S-transferase 5-5 in Salmonella typhimurium TA1535 leads to base-pair mutations upon exposure to dihalomethanes. | Thier, R., et al. 1993. Proc Natl Acad Sci U S A. 90: 8576-80. PMID: 8378332
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromomethyl Acetate, 5 g | sc-210964 | 5 g | $340.00 |
