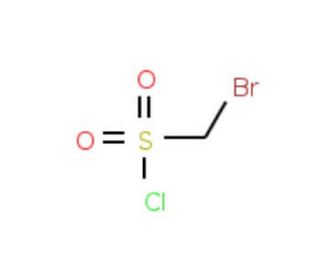
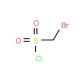
Bromomethanesulfonyl chloride (CAS 10099-08-8)
QUICK LINKS
Bromomethanesulfonyl chloride, an organosulfur compound, is a colorless liquid characterized by a strong odor. Its applications span a wide range, including organic synthesis, pharmaceuticals, and biochemistry. With its versatility, it serves as a valuable reagent for synthesizing various compounds, notably heterocycles, and can act as a catalyst in select reactions. Additionally, it plays a role as an intermediate in the production of certain drugs and chemicals. Extensively employed in scientific research, particularly within organic synthesis, pharmaceuticals, and biochemistry, bromomethanesulfonyl chloride holds significant utility. As an electrophilic reagent, it demonstrates a strong attraction to electron-rich molecules, while its nucleophilic properties enable it to react with electron-deficient molecules. Upon reaction with an electron-rich molecule, a covalent bond forms, resulting in the creation of a bromomethanesulfonate ester. Subsequently, this ester can participate in further reactions, contingent upon the characteristics of the involved molecule.
Bromomethanesulfonyl chloride (CAS 10099-08-8) References
- Synthesis and use of sulfonamide-, sulfoxide-, or sulfone-containing aminoglycoside-CoA bisubstrates as mechanistic probes for aminoglycoside N-6'-acetyltransferase. | Gao, F., et al. 2008. Bioorg Med Chem Lett. 18: 5518-22. PMID: 18805003
- Spiro[fluoreneisothiazolidin]one dioxides: new aldose reductase and L-hexonate dehydrogenase inhibitors. | DuPriest, MT., et al. 1991. J Med Chem. 34: 3229-34. PMID: 1956041
- Novel carvedilol analogues that suppress store-overload-induced Ca2+ release. | Smith, CD., et al. 2013. J Med Chem. 56: 8626-55. PMID: 24124794
- Reactive 5'-substituted thymidine derivatives as potential inhibitors of nucleotide biosynthesis. | Elliott, RD., et al. 1986. J Med Chem. 29: 1052-6. PMID: 2423689
- Protocol for Stereoselective Construction of Highly Functionalized Dienyl Sulfonyl Fluoride Warheads. | Zhang, ZW., et al. 2020. J Org Chem. 85: 13721-13734. PMID: 32960604
- Discovery of TAK-041: a Potent and Selective GPR139 Agonist Explored for the Treatment of Negative Symptoms Associated with Schizophrenia. | Reichard, HA., et al. 2021. J Med Chem. 64: 11527-11542. PMID: 34260228
- New antifilarial agents. 1. Epoxy sulfonamides and ethynesulfonamides. | Brienne, MJ., et al. 1987. J Med Chem. 30: 2232-9. PMID: 3681893
- Reactions of sulfenes with ketene acetals and ketene animals. | Truce, WE., et al. 1967. J Org Chem. 32: 990-7. PMID: 6042165
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromomethanesulfonyl chloride, 250 mg | sc-353113 | 250 mg | $821.00 | |||
Bromomethanesulfonyl chloride, 1 g | sc-353113A | 1 g | $1637.00 |
