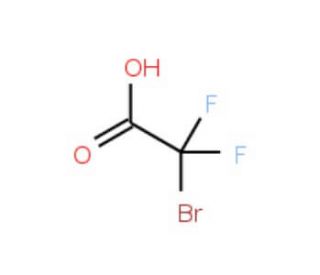
Bromodifluoroacetic acid (CAS 354-08-5)
QUICK LINKS
Bromodifluoroacetic acid (BDFA) is an organic compound that has gained widespread recognition in scientific research due to its remarkable versatility and effectiveness. It has been employed to investigate enzyme kinetics, unravel the intricacies of cell metabolism, and delve into the structure and function of proteins. Furthermore, Bromodifluoroacetic acid has been instrumental in studying the impact of environmental pollutants on aquatic organisms. Additionally, it has contributed to our understanding of the biosynthesis of specific hormones and neurotransmitters. As a halogenated organic compound, Bromodifluoroacetic acid possesses the ability to bind to specific molecules, thereby modifying their structure and function. This distinctive characteristic renders it an invaluable tool in exploring the mechanism of action of enzymes and proteins.
Bromodifluoroacetic acid (CAS 354-08-5) References
- Synthesis of 4,4-difluoroglycosides using ring-closing metathesis. | Audouard, C., et al. 2004. Org Biomol Chem. 2: 528-41. PMID: 14770232
- Nickel-Catalyzed Negishi Cross-Coupling of Bromodifluoroacetamides. | Tarui, A., et al. 2016. Org Lett. 18: 1128-31. PMID: 26910536
- Friedel-Crafts Fluoroacetylation of Indoles with Fluorinated Acetic Acids for the Synthesis of Fluoromethyl Indol-3-yl Ketones under Catalyst- and Additive-Free Conditions. | Yao, SJ., et al. 2016. J Org Chem. 81: 4226-34. PMID: 27101475
- Thermal conversion of primary alcohols to disulfides via xanthate intermediates: an extension to the Chugaev elimination. | He, W., et al. 2018. Org Biomol Chem. 16: 1659-1666. PMID: 29446434
- Catalytic One-Step Deoxytrifluoromethylation of Alcohols. | de Azambuja, F., et al. 2019. J Org Chem. 84: 2061-2071. PMID: 30632749
- Utilization of CO2 Feedstock for Organic Synthesis by Visible-Light Photoredox Catalysis. | Pradhan, S., et al. 2021. Chemistry. 27: 2254-2269. PMID: 32931070
- Late-stage difluoromethylation: concepts, developments and perspective. | Sap, JBI., et al. 2021. Chem Soc Rev. 50: 8214-8247. PMID: 34075979
- Pd-Catalyzed Difluoromethylations of Aryl Boronic Acids, Halides, and Pseudohalides with ICF2 H Generated ex Situ. | Gedde, OR., et al. 2022. Chemistry. 28: e202200997. PMID: 35388933
- Recent trends in dehydroxylative trifluoro-methylation, -methoxylation, -methylthiolation, and -methylselenylation of alcohols. | Cao, Y., et al. 2021. RSC Adv. 11: 39593-39606. PMID: 35492477
- C-H chlorination of (hetero)anilines via photo/organo co-catalysis. | Xie, W., et al. 2022. Org Biomol Chem. 20: 5319-5324. PMID: 35730736
- Catalytic and Stereoselective Transformations with Easily Accessible and Purchasable Allyl and Alkenyl Fluorides. | Paioti, PHS., et al. 2022. Angew Chem Int Ed Engl. 61: e202208742. PMID: 36017964
- One-pot synthesis of tri- and di-fluoromethylated bis(indolyl)methanols via Friedel-Crafts-type acylation and alkylation. | Wu, XT., et al. 2022. Org Biomol Chem. 20: 7491-7498. PMID: 36106547
- Visible-Light Promoted Radical Fluoroalkylation of O- and N-Substituted Alkenes. | Levin, VV. and Dilman, AD. 2023. Chem Rec. e202300038. PMID: 37017493
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromodifluoroacetic acid, 5 g | sc-285249 | 5 g | $38.00 | |||
Bromodifluoroacetic acid, 25 g | sc-285249A | 25 g | $142.00 |
