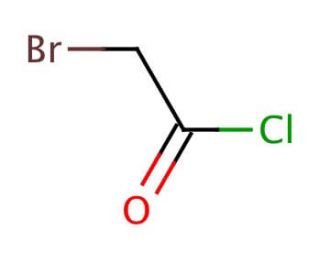
Molecular structure of Bromoacetyl chloride, CAS Number: 22118-09-8
Bromoacetyl chloride (CAS 22118-09-8)
Alternate Names:
2-Bromo-1-chloro-ethanal
Application:
Bromoacetyl chloride is used in the synthesis of α,α-disubstituted thioisomünchnones
CAS Number:
22118-09-8
Purity:
≥95%
Molecular Weight:
157.39
Molecular Formula:
C2H2BrClO
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Bromoacetyl chloride was used in the synthesis of alpha,alpha-disubstituted thioisomυnchnones. It was also used in the preparation of 1,3-dibromoacetone. It is a diphenyl ether that can be used to detect serine protease in tissue. It also has acylation activity and can be used in the synthesis of β-amino acids.
Bromoacetyl chloride (CAS 22118-09-8) References
- Nonadiabatic interactions in wave packet dynamics of the bromoacetyl chloride photodissociation. | Lasorne, B., et al. 2004. J Chem Phys. 120: 1271-8. PMID: 15268253
- Combined nonadiabatic transition-state theory and ab initio molecular dynamics study on selectivity of the alpha and beta bond fissions in photodissociation of bromoacetyl chloride. | Zhang, F., et al. 2006. J Chem Phys. 125: 184305. PMID: 17115750
- Nonadiabatic effects in C-Br bond scission in the photodissociation of bromoacetyl chloride. | Valero, R. and Truhlar, DG. 2006. J Chem Phys. 125: 194305. PMID: 17129101
- Competitive bond rupture in the photodissociation of bromoacetyl chloride and 2- and 3-bromopropionyl chloride: adiabatic versus diabatic dissociation. | Hsu, MY., et al. 2013. Chemphyschem. 14: 936-45. PMID: 23400968
- Rapid Fluorescent-Based Detection of New Delhi Metallo-β-Lactamases by Photo-Cross-Linking Using Conjugates of Azidonaphthalimide and Zinc(II)-Chelating Motifs. | Singha, M., et al. 2019. ACS Omega. 4: 10891-10898. PMID: 31460186
- Synthesis of Menthol Glycinates and Their Potential as Cooling Agents. | Klumpp, DA., et al. 2020. ACS Omega. 5: 4043-4049. PMID: 32149231
- From PARP1 to TNKS2 Inhibition: A Structure-Based Approach. | Tomassi, S., et al. 2020. ACS Med Chem Lett. 11: 862-868. PMID: 32435397
- Synthesis and Antiproliferative Evaluation of 3-Chloroazetidin-2-ones with Antimitotic Activity: Heterocyclic Bridged Analogues of Combretastatin A-4. | Malebari, AM., et al. 2021. Pharmaceuticals (Basel). 14: PMID: 34832901
- A complementary chemical probe approach towards customized studies of G-quadruplex DNA structures in live cells. | Prasad, B., et al. 2022. Chem Sci. 13: 2347-2354. PMID: 35310480
- Diels-Alder Adducts of Morphinan-6,8-Dienes and Their Transformations. | Marton, J., et al. 2022. Molecules. 27: PMID: 35566212
- Inhibitors of glucosamine-6-phosphate synthase as potential antimicrobials or antidiabetics - synthesis and properties. | Stefaniak, J., et al. 2022. J Enzyme Inhib Med Chem. 37: 1928-1956. PMID: 35801410
- Synthesis of 2,5-Dialkyl-1,3,4-oxadiazoles Bearing Carboxymethylamino Groups. | Łuczyński, M., et al. 2022. Molecules. 27: PMID: 36431787
- N-Acylated Ciprofloxacin Derivatives: Synthesis and In Vitro Biological Evaluation as Antibacterial and Anticancer Agents. | Struga, M., et al. 2023. ACS Omega. 8: 18663-18684. PMID: 37273589
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromoacetyl chloride, 25 ml | sc-227524 | 25 ml | $100.00 |
