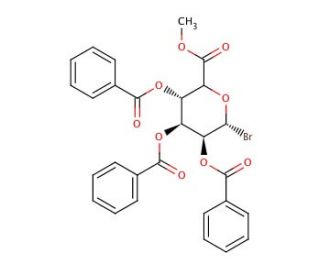
Bromo-2,3,4-tri-O-benzoyl-α-D-glucuronic Acid Methyl Ester (CAS 103674-69-7)
QUICK LINKS
Bromo-2,3,4-tri-O-benzoyl-α-D-glucuronic Acid Methyl Ester is a chemically synthesized ester of glucuronic acid prominently used in carbohydrate chemistry for structural and functional studies related to glucuronides. Its molecular structure, characterized by the presence of a bromo substituent and multiple benzoyl groups, enhances its reactivity, making it a valuable tool in glycoscience research. The bromine atom in particular introduces a reactive site, facilitating further derivatization or coupling reactions, which are fundamental in synthesizing more complex carbohydrate-based structures. This compound is extensively employed in the study of glucuronidation processes, a key pathway for the biotransformation of many small organic molecules in biological systems. By mimicking the glucuronic acid component of glucuronides, it serves as a precursor or intermediate in the synthesis of potential glycosidic linkages, aiding in the exploration of enzyme-substrate interactions within the UDP-glucuronosyltransferase (UGT) family. Researchers utilize this ester to probe the specificity and mechanism of UGT enzymes, which are crucial for understanding the metabolism of xenobiotics and endogenous substances without focusing on pharmacological aspects. Thus, it is instrumental in advancing fundamental research in enzymology and carbohydrate chemistry.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromo-2,3,4-tri-O-benzoyl-α-D-glucuronic Acid Methyl Ester, 200 mg | sc-207378 | 200 mg | $320.00 |
