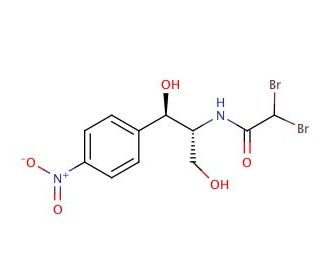
Bromamphenicol (CAS 16803-75-1)
QUICK LINKS
Bromamphenicol, indexed under CAS number 16803-75-1, is a brominated analog of the well-known compound chloramphenicol. Similar to its parent compound, bromamphenicol functions by binding to the 50S subunit of bacterial ribosomes, but it features a bromine atom replacing one of the hydrogens in the nitrobenzene ring. This subtle chemical modification influences its interaction with the bacterial ribosomal machinery. Bromamphenicol′s primary mode of action involves the inhibition of the peptidyl transferase activity, an essential enzymatic process in the synthesis of proteins. By interfering with this enzyme, bromamphenicol effectively blocks the formation of peptide bonds between amino acids, thereby inhibiting protein synthesis and stalling bacterial growth. In research, bromamphenicol has been used as a molecular tool to investigate the effects of halogenation on the pharmacodynamics of antibiotics. Studies have explored how such modifications impact the binding affinity and specificity of these molecules towards bacterial ribosomes, contributing to the broader field of antibiotic design. Research involving bromamphenicol helps in understanding the structural and functional dynamics of ribosomal inhibition, paving the way for the development of novel antibiotics and the enhancement of existing ones to counteract bacterial resistance more effectively.
Bromamphenicol (CAS 16803-75-1) References
- Studies on the mechanism of action of chloramphenicol. I. The conformation of chlioramphenicol in solution. | JARDETZKY, O. 1963. J Biol Chem. 238: 2498-508. PMID: 13957484
- A structural study of the interaction between the Dr haemagglutinin DraE and derivatives of chloramphenicol. | Pettigrew, DM., et al. 2009. Acta Crystallogr D Biol Crystallogr. 65: 513-22. PMID: 19465765
- Affinity labeling of the virginiamycin S binding site on bacterial ribosome. | Di Giambattista, M., et al. 1990. Biochemistry. 29: 9203-11. PMID: 2125475
- Mapping of Escherichia coli ribosomal components involved in peptidyl transferase activity. | Sonenberg, N., et al. 1973. Proc Natl Acad Sci U S A. 70: 1423-6. PMID: 4576018
- Some correlations between ribosomal proteins and ribosomal activities. | Zamir, A., et al. 1974. Adv Exp Med Biol. 44: 123-39. PMID: 4837682
- On the mechanism of action of chloramphenicol in protein synthesis. | Coutsogeorgopoulos, C. 1966. Biochim Biophys Acta. 129: 214-7. PMID: 5339107
- X-ray analysis of some antibiotic substances. | Dunitz, et al. 1950. Journal of the American Chemical Society. 72.9: 4276-4277.
- The crystal structure of chloramphenicol1 and bromamphenicol2. | Dunitz and J. D. 1952. Journal of the American Chemical Society. 74.4: 995-999.
- [83] Analogs of chloramphenicol and their application to labeling ribosomes. | Sonenberg, et al. 1977. Methods in Enzymology. Vol. 46, Academic Press: 703-707.
- 1H, 13C MAS NMR and GIAO-CPHF calculations of chloramphenicol, thiamphenicol and their pyrrole analogues. | Żołek and Teresa, et al. 2003. Journal of Molecular Structure. 646.1-3: 141-149.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromamphenicol, 5 mg | sc-391758 | 5 mg | $94.00 | |||
Bromamphenicol, 25 mg | sc-391758A | 25 mg | $328.00 |
