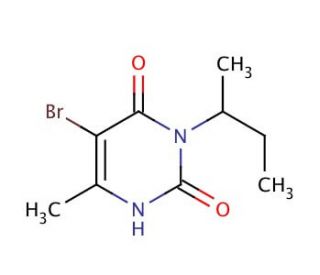
Molecular structure of Bromacil, CAS Number: 314-40-9
Bromacil (CAS 314-40-9)
CAS Number:
314-40-9
Molecular Weight:
261.12
Molecular Formula:
C9H13BrN2O2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Bromacil is an active ingredient extensively utilized for weed control in citrus orchards. This compound exists as a colorless to white crystalline solid without any discernible odor. Functioning as an herbicide, it is commercially available in the form of a wettable powder or in liquid formulations (NIOSH, 2016).
Bromacil (CAS 314-40-9) References
- Movement of bromacil and hexazinone in soils of Hawaiian pineapple fields. | Zhu, Y. and Li, QX. 2002. Chemosphere. 49: 669-74. PMID: 12430654
- Atrazine, bromacil, and diuron resistance in chlamydomonas: a single non-mendelian genetic locus controls the structure of the thylakoid binding site. | Galloway, RE. and Mets, LJ. 1984. Plant Physiol. 74: 469-74. PMID: 16663446
- Movement of bromacil in a Hawaii soil under pineapple cultivation - a field study. | Alavi, G., et al. 2008. Chemosphere. 72: 45-52. PMID: 18374966
- Effect of the adsorbate (Bromacil) equilibrium concentration in water on its adsorption on powdered activated carbon. Part 3: Competition with natural organic matter. | Al Mardini, F. and Legube, B. 2010. J Hazard Mater. 182: 10-7. PMID: 20619963
- Use of quadrupole time-of-flight mass spectrometry to determine proposed structures of transformation products of the herbicide bromacil after water chlorination. | Ibáñez, M., et al. 2011. Rapid Commun Mass Spectrom. 25: 3103-13. PMID: 21953966
- Photo-catalysis of bromacil under simulated solar light using Au/TiO₂: evaluation of main degradation products and toxicity implications. | Angthararuk, D., et al. 2015. Environ Sci Pollut Res Int. 22: 1468-79. PMID: 25163558
- In vitro selection of a single-stranded DNA molecular recognition element specific for bromacil. | Williams, RM., et al. 2014. J Nucleic Acids. 2014: 102968. PMID: 25400940
- Phytotoxicity of Alachlor, Bromacil and Diuron as single or mixed herbicides applied to wheat, melon, and molokhia. | El-Nahhal, Y. and Hamdona, N. 2015. Springerplus. 4: 367. PMID: 26207198
- Phytotoxicity of chiral herbicide bromacil: Enantioselectivity of photosynthesis in Arabidopsis thaliana. | Chen, Z., et al. 2016. Sci Total Environ. 548-549: 139-147. PMID: 26802342
- Degradation of bromacil by a Pseudomonas sp. | Chaudhry, GR. and Cortez, L. 1988. Appl Environ Microbiol. 54: 2203-7. PMID: 3056270
- Photoelectrocatalyzed degradation of a pesticides mixture solution (chlorfenvinphos and bromacil) by WO3 nanosheets. | Roselló-Márquez, G., et al. 2019. Sci Total Environ. 674: 88-95. PMID: 31004907
- Tolerance and sorption of Bromacil and Paraquat by thermophilic cyanobacteria Leptolyngbya 7M from Costa Rican thermal springs. | Brenes-Guillén, L., et al. 2019. J Contam Hydrol. 226: 103539. PMID: 31408829
- In-situ treatment of herbicide-contaminated groundwater-Feasibility study for the cases atrazine and bromacil using two novel nanoremediation-type materials. | Gawel, A., et al. 2020. J Hazard Mater. 393: 122470. PMID: 32208331
- Degradation of atrazine and bromacil in two forestry waste products. | James, TK., et al. 2021. Sci Rep. 11: 3284. PMID: 33558630
- Numerical Modeling of Groundwater Pollution by Chlorpyrifos, Bromacil and Terbuthylazine. Application to the Buñol-Cheste Aquifer (Spain). | Pérez-Indoval, R., et al. 2021. Int J Environ Res Public Health. 18: PMID: 33800654
Inhibitor of:
Atrazine.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bromacil, 250 mg | sc-257186 | 250 mg | $41.00 |
