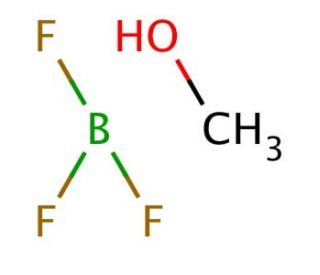
Boron trifluoride-methanol-complex solution (CAS 2802-68-8)
QUICK LINKS
Boron trifluoride-methanol-complex solution is utilized as a Lewis acid catalyst in various chemical reactions. It functions by coordinating with electron-rich species, activating them for nucleophilic attack or facilitating the formation of new carbon-carbon or carbon-heteroatom bonds. The complex solution acts by accepting a pair of electrons from the reactant, leading to the formation of a new bond and subsequent product formation. This interaction occurs at the molecular level, where the boron trifluoride component of the complex acts as a strong electrophile, attracting electron density from the methanol molecule. Its mechanism of action involves the activation of substrates through the formation of stable intermediates, allowing for the efficient and selective formation of desired products in various experimental applications.
Boron trifluoride-methanol-complex solution (CAS 2802-68-8) References
- Aflatoxin and ochratoxin A contamination of retail foods and intake of these mycotoxins in Japan. | Kumagai, S., et al. 2008. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 25: 1101-6. PMID: 19238621
- Extraction and enrichment of n-3 polyunsaturated fatty acids and ethyl esters through reversible π-π complexation with aromatic rings containing ionic liquids. | Cheong, LZ., et al. 2011. J Agric Food Chem. 59: 8961-7. PMID: 21790198
- Effect of prolonged refrigeration on the lipid profile, lipase activity, and oxidative status of human milk. | Bertino, E., et al. 2013. J Pediatr Gastroenterol Nutr. 56: 390-6. PMID: 23149807
- Docosahexaenoic acid in maternal and neonatal plasma phospholipids and milk lipids of Taiwanese women in Kinmen: fatty acid composition of maternal blood, neonatal blood and breast milk. | Huang, HL., et al. 2013. Lipids Health Dis. 12: 27. PMID: 23496882
- Comparative analysis of EPA and DHA in fish oil nutritional capsules by GC-MS. | Yi, T., et al. 2014. Lipids Health Dis. 13: 190. PMID: 25496531
- Method for determination of fatty acids in bovine colostrum using GC-FID. | Yurchenko, S., et al. 2016. Food Chem. 212: 117-22. PMID: 27374514
- Preparation and characterization of (10)B boric acid with high purity for nuclear industry. | Zhang, W., et al. 2016. Springerplus. 5: 1202. PMID: 27516940
- Fatty acid profiling of 75 Indian snack samples highlights overall low trans fatty acid content with high polyunsaturated fatty acid content in some samples. | Joshee, K., et al. 2019. PLoS One. 14: e0225798. PMID: 31805103
- Production of feruloylated lysophospholipids via a one-step enzymatic interesterification. | Rychlicka, M., et al. 2020. Food Chem. 323: 126802. PMID: 32311619
- Nutritive Value of 11 Bee Pollen Samples from Major Floral Sources in Taiwan. | Hsu, PS., et al. 2021. Foods. 10: PMID: 34574339
- Dietary Phospholipids Prepared From Scallop Internal Organs Attenuate the Serum and Liver Cholesterol Contents by Enhancing the Expression of Cholesterol Hydroxylase in the Liver of Mice. | Sugimoto, K., et al. 2021. Front Nutr. 8: 761928. PMID: 34778346
- The Importance of Fatty Acid Precision Nutrition: Effects of Dietary Fatty Acid Composition on Growth, Hepatic Metabolite, and Intestinal Microbiota in Marine Teleost Trachinotus ovatus. | Zhang, G., et al. 2023. Aquac Nutr. 2023: 2556799. PMID: 36860978
- Variability of fatty acid content in pumpkin seeds (Cucurbita pepo L.). | Murkovic, M., et al. 1996. Z Lebensm Unters Forsch. 203: 216-9. PMID: 8873459
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boron trifluoride-methanol-complex solution, 500 ml | sc-234204 | 500 ml | $80.00 | |||
Boron trifluoride-methanol-complex solution, 1 L | sc-234204A | 1 L | $110.00 |
