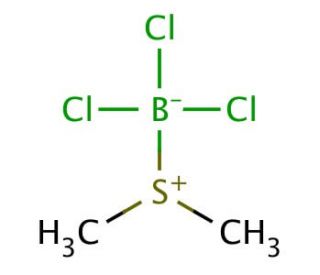
Molecular structure of Boron trichloride methyl sulfide complex, CAS Number: 5523-19-3
Boron trichloride methyl sulfide complex (CAS 5523-19-3)
Application:
Boron trichloride methyl sulfide complex is a Boron trichloride in convenient solid form
CAS Number:
5523-19-3
Molecular Weight:
179.30
Molecular Formula:
C2H6BCl3S
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Boron trichloride methyl sulfide complex (BTMSC) is a coordination compound supplied in a convenient solid form. It is used in the synthesis of organic compounds, as a catalyst in chemical reactions, and as a reagent in biochemical and physiological studies. In organic synthesis, Boron trichloride methyl sulfide complex is employed to catalyze the reaction of aldehydes and ketones with organometallic reagents. In addition, the complex interacts with various biomolecules, such as proteins, to modulate their activity.
Boron trichloride methyl sulfide complex (CAS 5523-19-3) References
- Design, synthesis, and biological evaluation of 14-substituted aromathecins as topoisomerase I inhibitors. | Cinelli, MA., et al. 2008. J Med Chem. 51: 4609-19. PMID: 18630891
- A concise and straightforward total synthesis of (+/-)-salinosporamide A, based on a biosynthesis model. | Mulholland, NP., et al. 2008. Org Biomol Chem. 6: 2782-9. PMID: 18633536
- The structure-activity relationships of A-ring-substituted aromathecin topoisomerase I inhibitors strongly support a camptothecin-like binding mode. | Cinelli, MA., et al. 2010. Bioorg Med Chem. 18: 5535-52. PMID: 20630766
- Covalent molecular assembly: construction of ultrathin multilayer films by a two-dimensional fabrication method. | Zhou, R. and Srinivasan, MP. 2013. J Colloid Interface Sci. 392: 158-166. PMID: 23127877
- Optimization of GPR40 Agonists for Type 2 Diabetes. | Liu, JJ., et al. 2014. ACS Med Chem Lett. 5: 517-21. PMID: 24900872
- 4-Benzothiazole-7-hydroxyindolinyl diaryl ureas are potent P2Y1 antagonists with favorable pharmacokinetics: low clearance and small volume of distribution. | Qiao, JX., et al. 2014. ChemMedChem. 9: 2327-43. PMID: 24989964
- Synthesis of Homoverrucosanoid-Derived Esters and Evaluation as MDR Modulators. | Schäfer, A., et al. 2017. J Org Chem. 82: 10504-10522. PMID: 28949536
- Synthesis of medium ring ethers. 5. The synthesis of (+)-laurencin | Burton, J. W., Clark, J. S., Derrer, S., Stork, T. C., Bendall, J. G., & Holmes, A. B. 1997. Journal of the American Chemical Society. 119(32): 7483-7498.
- Total synthesis of the thiopeptide promothiocin A | Bagley, M. C., Bashford, K. E., Hesketh, C. L., & Moody, C. J. 2000. Journal of the American Chemical Society. 122(14): 3301-3313.
- Nitrone dipolar cycloaddition routes to piperidines and indolizidines. Part 9. Formal synthesis of (−)-pinidine and total synthesis of (−)-histrionicotoxin,(+)-histrionicotoxin and (−)-histrionicotoxin 235A | Davison, E. C., Fox, M. E., Holmes, A. B., Roughley, S. D., Smith, C. J., Williams, G. M.,.. & Thompson, M. J. 2002. Journal of the Chemical Society, Perkin Transactions 1. (12): 1494-1514.
- Synthesis of medium-ring lactones via tandem methylenation/Claisen rearrangement of cyclic carbonates | Anderson, E. A., Davidson, J. E., Harrison, J. R., T O'Sullivan, P., Burton, J. W., Collins, I., & Holmes, A. B. 2002. Tetrahedron. 58(10): 1943-1971.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boron trichloride methyl sulfide complex, 25 g | sc-252508 | 25 g | $195.00 |
