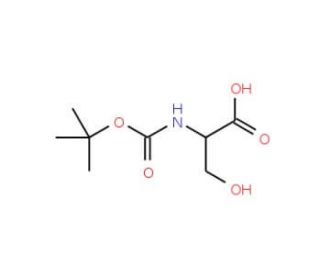
Boc-Ser-OH (CAS 3262-72-4)
QUICK LINKS
Boc-Ser-OH is a chemical compound that functions as a protecting group for the amino acid serine in peptide synthesis. It plays a role in the selective protection of the hydroxyl group of serine, allowing for specific reactions to occur at other functional groups within the peptide chain. The compound interacts at the molecular level by forming a covalent bond with the hydroxyl group of serine, thereby preventing unwanted side reactions during peptide synthesis. This protection allows for the manipulation of the peptide chain at other sites without affecting the serine residue. Boc-Ser-OH′s mechanism of action involves the reversible attachment of the Boc group to the hydroxyl group of serine, providing a temporary shield during chemical reactions. Boc-Ser-Oh′s function is to facilitate the controlled assembly of peptides with specific sequences and structures.
Boc-Ser-OH (CAS 3262-72-4) References
- The synthesis, distribution, and anti-hepatic cancer activity of YSL. | Ding, W., et al. 2004. Bioorg Med Chem. 12: 4989-94. PMID: 15336278
- The 'O-acyl isopeptide method' for the synthesis of difficult sequence-containing peptides: application to the synthesis of Alzheimer's disease-related amyloid beta peptide (Abeta) 1-42. | Sohma, Y., et al. 2005. J Pept Sci. 11: 441-51. PMID: 15761877
- 2-(N-Fmoc)-3-(N-Boc-N-methoxy)-diaminopropanoic acid, an amino acid for the synthesis of mimics of O-linked glycopeptides. | Carrasco, MR., et al. 2006. Biopolymers. 84: 414-20. PMID: 16508952
- Synthesis of amyloid beta peptide 1-42 (E22Delta) click peptide: pH-triggered in situ production of its native form. | Wang, H., et al. 2009. Bioorg Med Chem. 17: 4881-7. PMID: 19564119
- Facile Synthesis of Boc-Protected Selenocystine and its Compatibility with Late-Stage Farnesylation at Cysteine Site. | Zhu, PC. and Chen, YX. 2021. Protein Pept Lett. 28: 603-611. PMID: 33357178
- Double-drug loading upconversion nanoparticles for monitoring and therapy of a MYC/BCL6-positive double-hit diffuse large B-cell lymphoma. | Yang, Y., et al. 2022. Biomaterials. 287: 121607. PMID: 35696785
- Cyclols, cyclodepsipeptides, and N-acyl-diketopiperazines from linear precursors. Synthesis and crystal structure of 10-membered cyclodepsipeptides. | Zanotti, G., et al. 1983. Int J Pept Protein Res. 22: 410-21. PMID: 6654588
- Preparation of an aspartic acid-containing protected peptide. Alternatives for overcoming a side reaction during HF treatment. | Robles, J., et al. 1994. Int J Pept Protein Res. 43: 359-62. PMID: 8045681
- [18] Synthesis of O-phosphoserine- and O-phosphothreonine-containing peptides | John W. Perich. 1991. Methods in Enzymology. 201: 225-233.
- Cyclohexyl ether as a new hydroxy-protecting group for serine in solid-phase peptide synthesis | Yasuhiro Nishiyama, Keisuke Kurita. 1999. Tetrahedron Letters. 40: 927-930.
- Cyclohexyl ether as a new hydroxy-protecting group for serine and threonine in peptide synthesis | Yasuhiro Nishiyama,†*a Suguru Shikama,a Ken-ichi Moritaa and Keisuke Kuritaa. 2000. Chem. Soc., Perkin Trans. 1: 1949-1954.
- (Z)-Alkene phospho-Ser-cis-Pro substrate analog for Pin1, a phosphorylation-dependent peptidyl-prolyl isomerase | Scott A. Hart & Felicia A. Etzkorn. 2002. Peptides for the New Millennium. 478–480.
- Design and synthesis of a novel water-soluble Aβ1-42 isopeptide: an efficient strategy for the preparation of Alzheimer's disease-related peptide, Aβ1-42, via O–N intramolecular acyl migration reaction | Youhei Sohma, Masato Sasaki, Yoshio Hayashi, Tooru Kimura, Yoshiaki Kiso. 2004. Tetrahedron Letters. 45: 5965-5968.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boc-Ser-OH, 5 g | sc-252502 | 5 g | $19.00 |
