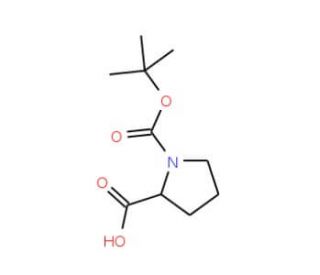
Molecular structure of Boc-Pro-OH, CAS Number: 15761-39-4
Boc-Pro-OH (CAS 15761-39-4)
Alternate Names:
Boc-L-proline; N-(tert-Butoxycarbonyl)-L-proline
Application:
Boc-Pro-OH is a useful synthetic intermediate
CAS Number:
15761-39-4
Molecular Weight:
215.25
Molecular Formula:
C10H17NO4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Boc-Pro-OH, a white crystalline powder soluble in ethanol and water, is a derivative of proline, a non-essential amino acid vital for protein biosynthesis. It is extensively applied in a variety of scientific research, especially in the synthesis of peptides, serving as a common protecting group for the amine groups of amino acids during such synthesis. Additionally, Boc-Pro-OH is used as a chiral auxiliary in asymmetric synthesis, underlining its multifaceted applications in scientific experiments. Boc-Pro-OH is a useful synthetic intermediate. It is used to synthesize Daclatasvir which inhibits the HCV protein NS5A.
Boc-Pro-OH (CAS 15761-39-4) References
- Visible-Light-Mediated Decarboxylative Radical Additions to Vinyl Boronic Esters: Rapid Access to γ-Amino Boronic Esters. | Noble, A., et al. 2018. Angew Chem Int Ed Engl. 57: 2155-2159. PMID: 29316095
- Thiocarbonyl-enabled ferrocene C-H nitrogenation by cobalt(III) catalysis: thermal and mechanochemical. | Yetra, SR., et al. 2018. Beilstein J Org Chem. 14: 1546-1553. PMID: 30013681
- Regioselective Hydroalkylation and Arylalkylation of Alkynes by Photoredox/Nickel Dual Catalysis: Application and Mechanism. | Yue, H., et al. 2020. Angew Chem Int Ed Engl. 59: 5738-5746. PMID: 31901214
- Discovery of Kynurenines Containing Oligopeptides as Potent Opioid Receptor Agonists. | Szűcs, E., et al. 2020. Biomolecules. 10: PMID: 32059524
- Imidazopyridine-fused [1,3]diazepinones: modulations of positions 2 to 4 and their impacts on the anti-melanoma activity. | Baccon-Sollier, PL., et al. 2020. J Enzyme Inhib Med Chem. 35: 935-949. PMID: 32249633
- Altering the Sex Pheromone Cyclo(l-Pro-l-Pro) of the Diatom Seminavis robusta towards a Chemical Probe. | Bonneure, E., et al. 2021. Int J Mol Sci. 22: PMID: 33494376
- Rapid Optimization of Photoredox Reactions for Continuous-Flow Systems Using Microscale Batch Technology. | González-Esguevillas, M., et al. 2021. ACS Cent Sci. 7: 1126-1134. PMID: 34345665
- An efficient one-pot conversion of carboxylic acids into benzimidazoles via an HBTU-promoted methodology. | Barasa, L. and Yoganathan, S. 2018. RSC Adv. 8: 35824-35830. PMID: 35547918
- The Anti-Tubercular Aminolipopeptide Trichoderin A Displays Selective Toxicity against Human Pancreatic Ductal Adenocarcinoma Cells Cultured under Glucose Starvation. | Kasim, JK., et al. 2023. Pharmaceutics. 15: PMID: 36678914
- Synthesis of Novel 1-Oxo-2,3,4-trisubstituted Tetrahydroisoquinoline Derivatives, Bearing Other Heterocyclic Moieties and Comparative Preliminary Study of Anti-Coronavirus Activity of Selected Compounds. | Kandinska, MI., et al. 2023. Molecules. 28: PMID: 36771170
- Organocatalysis with carbon nitrides. | Ruban, SM., et al. 2023. Sci Technol Adv Mater. 24: 2188879. PMID: 37007670
- Regioselective Cyclic Iminium Formation of Ugi Advanced Intermediates: Rapid Access to 3,4-Dihydropyrazin-2(1H)-ones and Other Diverse Nitrogen-Containing Heterocycles. | Cankařová, N. and Krchňák, V. 2023. Molecules. 28: PMID: 37049824
- The Parallel Structure-Activity Relationship Screening of Three Compounds Identifies the Common Agonist Pharmacophore of Pyrrolidine Bis-Cyclic Guanidine Melanocortin-3 Receptor (MC3R) Small-Molecule Ligands. | Ericson, MD., et al. 2023. Int J Mol Sci. 24: PMID: 37373293
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boc-Pro-OH, 5 g | sc-300292 | 5 g | $31.00 | |||
Boc-Pro-OH, 25 g | sc-300292A | 25 g | $84.00 |
