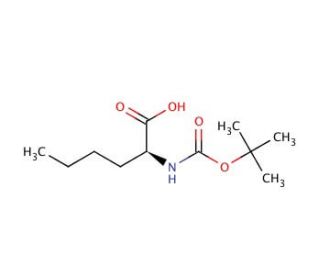
Molecular structure of Boc-Nle-OH, CAS Number: 6404-28-0
Boc-Nle-OH (CAS 6404-28-0)
Application:
Boc-Nle-OH is a boc protected amino acid derivative
CAS Number:
6404-28-0
Purity:
99%
Molecular Weight:
231.29
Molecular Formula:
C11H21NO4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Boc-Nle-OH is an amino acid derivative with wide-ranging applications in scientific research. Its versatility makes it valuable in studying various biochemical and physiological effects. Moreover, Boc-Nle-OH is an essential component in the synthesis of diverse compounds. Notably, researchers employ it to modify the structure and activity of peptides and proteins.
Boc-Nle-OH (CAS 6404-28-0) References
- Chemoenzymatic synthesis of neoglycopeptides using endo beta-N-acetylglucosaminidase from Mucor hiemalis. | Haneda, K., et al. 2003. Methods Enzymol. 362: 74-85. PMID: 12968358
- Synthesis and biological activity of CCK heptapeptide analogues. Effects of conformational constraints and standard modifications on receptor subtype selectivity, functional activity in vitro, and appetite suppression in vivo. | Holladay, MW., et al. 1992. J Med Chem. 35: 2919-28. PMID: 1501220
- Synthesis and evaluation of TAC-based inhibitors of papain as mimics of cystatin B. | van Zoelen, DJ., et al. 2007. Chembiochem. 8: 1950-6. PMID: 17886319
- Synthesis and binding affinities of cyclic and related linear analogues of CCK8 selective for central receptors. | Charpentier, B., et al. 1989. J Med Chem. 32: 1184-90. PMID: 2724293
- Application of a Sulfoxonium Ylide Electrophile to Generate Cathepsin X-Selective Activity-Based Probes. | Mountford, SJ., et al. 2020. ACS Chem Biol. 15: 718-727. PMID: 32022538
- Imidazopyridine-fused [1,3]diazepinones: modulations of positions 2 to 4 and their impacts on the anti-melanoma activity. | Baccon-Sollier, PL., et al. 2020. J Enzyme Inhib Med Chem. 35: 935-949. PMID: 32249633
- Pentagastrin analogs containing alpha-aminooxy acids, I. Synthesis of analogs substituted at the N-terminus. | Kisfaludy, L., et al. 1978. Hoppe Seylers Z Physiol Chem. 359: 887-95. PMID: 711151
- Design and synthesis of thrombin substrates with modified kinetic parameters. | Rijkers, DT., et al. 1995. Thromb Res. 79: 491-9. PMID: 7502275
- Synthesis and biological evaluation of cholecystokinin analogs in which the Asp-Phe-NH2 moiety has been replaced by a 3-amino-7-phenylheptanoic acid or a 3-amino-6-(phenyloxy)hexanoic acid. | Amblard, M., et al. 1993. J Med Chem. 36: 3021-8. PMID: 7692048
- Pegylated peptides. II. Solid-phase synthesis of amino-, carboxy- and side-chain pegylated peptides. | Lu, YA. and Felix, AM. 1994. Int J Pept Protein Res. 43: 127-38. PMID: 8200730
- Synthesis of lipidic amino acid and dipeptide inhibitors of human platelet phospholipase A2. | Kokotos, G., et al. 1996. Int J Pept Protein Res. 48: 160-6. PMID: 8872534
- Synthesis and biological evaluation of potent, selective, hexapeptide CCK-A agonist anorectic agents. | Pierson, ME., et al. 1997. J Med Chem. 40: 4302-7. PMID: 9435899
- Synthesis and conformational studies of proline-containing tripeptides | AM Tamburro, V Guantieri, A Scatturin - International Journal of Biological …, 1984 - Elsevier. October 1984,. International Journal of Biological Macromolecules. Volume 6, Issue 5,: Pages 241-248.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boc-Nle-OH, 1 g | sc-239400 | 1 g | $80.00 |
