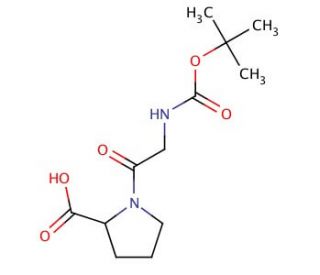
Boc-Gly-Pro-OH (CAS 14296-92-5)
QUICK LINKS
Boc-Gly-Pro-OH, a peptide compound, has emerged as a valuable tool in scientific research due to its versatility and wide-ranging applications in peptide synthesis and biochemical studies. One significant aspect of its mechanism of action lies in its role as a protected amino acid derivative, specifically featuring a tert-butyloxycarbonyl (Boc) protecting group attached to the amino group of glycine. This protecting group serves to shield the amino functionality during peptide synthesis, allowing for selective deprotection and subsequent coupling reactions with other amino acids. Boc-Gly-Pro-OH has been extensively utilized in solid-phase peptide synthesis (SPPS) as a building block for the assembly of peptide chains with precise control over sequence and length. Moreover, researchers have employed Boc-Gly-Pro-OH as a substrate in enzymatic assays to investigate the specificity and activity of various peptidases and proteases. Additionally, structural modifications of Boc-Gly-Pro-OH have been explored to enhance its stability, bioavailability, and affinity towards target molecules, facilitating the development molecular probes. Overall, Boc-Gly-Pro-OH remains a fundamental component in peptide chemistry and biochemical research, contributing to the advancement of peptide-based technologies and understanding of biological processes.
Boc-Gly-Pro-OH (CAS 14296-92-5) References
- Synthesis and use of 4-peptidylhydrazido-N-hexyl-1,8-naphthalimides as fluorogenic histochemical substrates for dipeptidyl peptidase IV and tripeptidyl peptidase I. | Ivanov, I., et al. 2009. Eur J Med Chem. 44: 384-92. PMID: 18410982
- Ferrocene tripeptide Gly-Pro-Arg conjugates: synthesis and inhibitory effects on Alzheimer's Aβ(1-42) fibrillogenesis and Aβ-induced cytotoxicity in vitro. | Zhou, B., et al. 2013. Bioorg Med Chem. 21: 395-402. PMID: 23245572
- Bioluminescent Probe for Monitoring Endogenous Fibroblast Activation Protein-Alpha. | Lin, Y., et al. 2019. Anal Chem. 91: 14873-14878. PMID: 31670506
- Synthetic identification as a hexapeptide of alpha substance-IB inducing sexual agglutination in Saccharomyces cerevisiae. | Aoyagi, H., et al. 1977. Experientia. 33: 870-1. PMID: 330197
- A Novel Chemiluminescence Probe for Sensitive Detection of Fibroblast Activation Protein-Alpha In Vitro and in Living Systems. | Fu, A., et al. 2021. Anal Chem. 93: 6501-6507. PMID: 33866786
- Molecular and in vivo studies of a glutamate-class prolyl-endopeptidase for coeliac disease therapy. | Del Amo-Maestro, L., et al. 2022. Nat Commun. 13: 4446. PMID: 35915115
- Isoperoxidase spectra of single tulip cultivars and their parrot mutants. | Saniewski, M. and Puchalski, J. 1977. Experientia. 33: 869-70. PMID: 891757
- Collagen-based structures containing the peptoid residue N-isobutylglycine (Nleu): synthesis and biophysical studies of Gly-Pro-Nleu sequences by circular dichroism, ultraviolet absorbance, and optical rotation. | Feng, Y., et al. 1996. Biopolymers. 39: 859-72. PMID: 8946805
- Steric effects of cis‐trans isomerism on neighboring residues in proline oligopeptides: A 13C‐nmr study of conformational heterogeneity in linear tripeptides | Deslauriers, R., et al. 1979. Biopolymers: Original Research on Biomolecules. 18(3): 523-538.
- Oxidation of di-peptides with Mn (III): Synthesis, characterization and mechanistic study | Kumara, M. N., et al. 2001. Reaction Kinetics and Catalysis Letters. 72: 331-342.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boc-Gly-Pro-OH, 5 g | sc-285154 | 5 g | $165.00 | |||
Boc-Gly-Pro-OH, 25 g | sc-285154A | 25 g | $693.00 |
