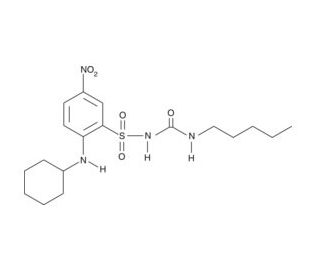
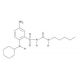
BM 567 (CAS 284464-77-3)
See product citations (1)
QUICK LINKS
BM 567, identified by CAS number 284464-77-3, is a chemical that has been extensively utilized in research settings, particularly for its role as a selective antagonist of specific receptors within the endocrine system. The compound′s mechanism of action revolves around its ability to bind to and inhibit the activity of estrogen receptors, particularly the subtype known as ERβ (estrogen receptor beta). This selective antagonism is critical for studies aimed at deciphering the distinct roles of various estrogen receptors in cellular and molecular processes. By blocking ERβ, BM 567 allows researchers to isolate and investigate the unique contributions of this receptor in various physiological and biochemical pathways, independent of ERα (estrogen receptor alpha) activities. This has proven invaluable in the field of endocrine research, where understanding the differential roles of receptor subtypes can explain their individual contributions to cellular regulation and the modulation of gene expression. BM 567′s use in experimental models has facilitated deeper insights into the receptor-specific actions that underpin multiple regulatory systems within the body, providing a clearer view of the molecular underpinnings that guide receptor-mediated signaling and response in various tissues.
BM 567 (CAS 284464-77-3) References
- Structure determination and comparison of BM567, a sulfonylurea, with terbogrel, two compounds with dual action, thromboxane receptor antagonism and thromboxane synthase inhibition. | Michaux, C., et al. 2001. Bioorg Med Chem Lett. 11: 1019-22. PMID: 11327579
- Pharmacological evaluation of the novel thromboxane modulator BM-567 (I/II). Effects of BM-567 on platelet function. | Dogné, JM., et al. 2003. Prostaglandins Leukot Essent Fatty Acids. 68: 49-54. PMID: 12538090
- Pharmacological evaluation of the novel thromboxane modulator BM-567 (II/II). Effects of BM-567 on osteogenic sarcoma-cell-induced platelet aggregation. | de Leval, X., et al. 2003. Prostaglandins Leukot Essent Fatty Acids. 68: 55-9. PMID: 12538091
- Evaluation of original dual thromboxane A2 modulators as antiangiogenic agents. | de Leval, X., et al. 2006. J Pharmacol Exp Ther. 318: 1057-67. PMID: 16720756
- Isoprostanes inhibit vascular endothelial growth factor-induced endothelial cell migration, tube formation, and cardiac vessel sprouting in vitro, as well as angiogenesis in vivo via activation of the thromboxane A(2) receptor: a potential link between oxidative stress and impaired angiogenesis. | Benndorf, RA., et al. 2008. Circ Res. 103: 1037-46. PMID: 18802021
- Involvement of the thromboxane A2 receptor in the regulation of steroidogenic acute regulatory gene expression in murine Leydig cells. | Pandey, AK., et al. 2009. Endocrinology. 150: 3267-73. PMID: 19325001
- Kupffer cell depletion attenuates leptin-mediated methoxamine-stimulated portal perfusion pressure and thromboxane A2 release in a rodent model of NASH-cirrhosis. | Yang, YY., et al. 2012. Clin Sci (Lond). 123: 669-80. PMID: 22734967
- Determination of role of thromboxane A2 in rheumatoid arthritis. | Wang, MJ., et al. 2015. Discov Med. 19: 23-32. PMID: 25636958
- Human cytochrome P450 enzymes 5-51 as targets of drugs and natural and environmental compounds: mechanisms, induction, and inhibition - toxic effects and benefits. | Rendic, SP. and Peter Guengerich, F. 2018. Drug Metab Rev. 50: 256-342. PMID: 30717606
- Anatomy and size of Megateuthis, the largest belemnite. | Klug, C., et al. 2024. Swiss J Palaeontol. 143: 23. PMID: 38827169
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
BM 567, 1 mg | sc-205229 | 1 mg | $72.00 | |||
BM 567, 5 mg | sc-205229A | 5 mg | $435.00 |
