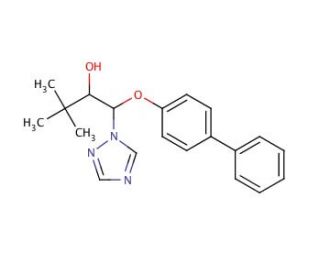
Bitertanol (CAS 55179-31-2)
See product citations (1)
QUICK LINKS
Bitertanol is a chemical used primarily in the field of agricultural research for its fungicidal properties. It is a triazole compound that inhibits the biosynthesis of ergosterol, an essential component of fungal cell membranes, providing useful for studying fungal growth and proliferation. Researchers employ bitertanol to understand the resistance mechanisms that fungi can develop against fungicides, which is critical for the management of plant diseases. Studies involving bitertanol also focus on its effects on various pathogenic fungi that affect crops, contributing to the development of strategies for crop protection and yield improvement. Moreover, research is conducted on the environmental impact of bitertanol, including its persistence in soil and water, and its potential effects on non-target organisms, which is important for assessing its ecological safety.
Bitertanol (CAS 55179-31-2) References
- Determination of tridemorph and other fungicide residues in fruit samples by liquid chromatography-electrospray tandem mass spectrometry. | Zamora, T., et al. 2004. J Chromatogr A. 1045: 137-43. PMID: 15378888
- Induction and inhibition of cytochrome P450-dependent monooxygenases of rats by fungicide bitertanol. | Chan, PK., et al. 2006. Food Chem Toxicol. 44: 2047-57. PMID: 16971034
- Kinetic aspects and identification of by-products during the ozonation of bitertanol in agricultural wastewaters. | Bourgin, M., et al. 2013. Chemosphere. 90: 1387-95. PMID: 23000045
- Currently used pesticides and their mixtures affect the function of sex hormone receptors and aromatase enzyme activity. | Kjeldsen, LS., et al. 2013. Toxicol Appl Pharmacol. 272: 453-64. PMID: 23871939
- Direct enantioseparation of nitrogen-heterocyclic pesticides on cellulose-based chiral column by high-performance liquid chromatography. | Chai, T., et al. 2015. Chirality. 27: 32-8. PMID: 25331721
- Effects of currently used pesticides and their mixtures on the function of thyroid hormone and aryl hydrocarbon receptor in cell culture. | Ghisari, M., et al. 2015. Toxicol Appl Pharmacol. 284: 292-303. PMID: 25684042
- Stereoselective Separation of the Fungicide Bitertanol Stereoisomers by High-Performance Liquid Chromatography and Their Degradation in Cucumber. | Li, L., et al. 2018. J Agric Food Chem. 66: 13303-13309. PMID: 30495953
- Placental transfer of pesticides studied in human placental perfusion. | Mathiesen, L., et al. 2020. Basic Clin Pharmacol Toxicol. 127: 505-515. PMID: 32558230
- Stereoselective environmental behavior and biological effects of the chiral bitertanol. | Li, L., et al. 2020. Sci Total Environ. 728: 138867. PMID: 32570326
- Multiresidues of environmental contaminants in bats from Turkey. | Kuzukiran, O., et al. 2021. Chemosphere. 282: 131022. PMID: 34090000
- Pesticides in fruits and vegetables from the Souss Massa region, Morocco. | Choubbane, H., et al. 2022. Food Addit Contam Part B Surveill. 15: 79-88. PMID: 35076356
- Azole pesticide products and their hepatic metabolites cause endocrine disrupting potential by suppressing the homo-dimerization of human estrogen receptor alpha. | Jung, DW., et al. 2023. Environ Pollut. 318: 120894. PMID: 36549450
- Bitertanol, a triazole fungicide, increases operant responding but not motor activity. | Allen, AR. and MacPhail, RC. 1993. Neurotoxicol Teratol. 15: 237-42. PMID: 8413077
- Simple and rapid method for simultaneous gas chromatographic determination of bitertanol, metalaxyl, oxadixyl, propiconazole, and triadimefon residues in cucumbers. | Lee, WO. and Wong, SK. 1995. Analyst. 120: 2475-8. PMID: 8540615
- Determination of bitertanol residues in strawberries by liquid chromatography with fluorescence detection and confirmation by gas chromatography/mass spectrometry. | Yamazaki, Y. and Ninomiya, T. 1998. J AOAC Int. 81: 1252-6. PMID: 9850587
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bitertanol, 250 mg | sc-234101 | 250 mg | $40.00 |
