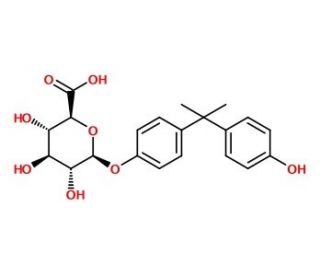
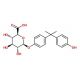
Bisphenol A β-D-Glucuronide (CAS 267244-08-6)
See product citations (4)
QUICK LINKS
Bisphenol A β-D-Glucuronide is a metabolite of Bisphenol A (BPA), a compound extensively studied for its environmental presence and potential biological effects. In research applications, this glucuronide form is particularly important for understanding the metabolic fate of BPA, as it represents a phase II conjugation product that facilitates the body′s elimination of BPA. Laboratories utilize Bisphenol A β-D-Glucuronide to investigate the detoxification processes that occur in organisms, shedding light on the metabolic pathways involved in the processing of xenobiotic compounds. Moreover, it serves as a reference standard in analytical studies that measure BPA exposure, using techniques such as liquid chromatography-mass spectrometry, to enable detection and quantification in biological samples.
Bisphenol A β-D-Glucuronide (CAS 267244-08-6) References
- In vitro and in vivo interactions of bisphenol A and its metabolite, bisphenol A glucuronide, with estrogen receptors alpha and beta. | Matthews, JB., et al. 2001. Chem Res Toxicol. 14: 149-57. PMID: 11258963
- Excretion of bisphenol A-glucuronide into the small intestine and deconjugation in the cecum of the rat. | Sakamoto, H., et al. 2002. Biochim Biophys Acta. 1573: 171-6. PMID: 12399027
- Interactions of phytoestrogens with estrogen receptors alpha and beta (III). Estrogenic activities of soy isoflavone aglycones and their metabolites isolated from human urine. | Kinjo, J., et al. 2004. Biol Pharm Bull. 27: 185-8. PMID: 14758030
- Genistein and daidzein induce cell proliferation and their metabolites cause oxidative DNA damage in relation to isoflavone-induced cancer of estrogen-sensitive organs. | Murata, M., et al. 2004. Biochemistry. 43: 2569-77. PMID: 14992594
- Inhibition of genistein glucuronidation by bisphenol A in human and rat liver microsomes. | Coughlin, JL., et al. 2012. Drug Metab Dispos. 40: 481-5. PMID: 22146138
- Bisphenol A-glucuronide measurement in urine samples. | Harthé, C., et al. 2012. Talanta. 100: 410-3. PMID: 23141357
- Development and validation of an LC-MS/MS method for simultaneous quantitative analysis of free and conjugated bisphenol A in human urine. | Battal, D., et al. 2014. Biomed Chromatogr. 28: 686-93. PMID: 24343900
- Quantitative determination of free and total bisphenol A in human urine using labeled BPA glucuronide and isotope dilution mass spectrometry. | Kubwabo, C., et al. 2014. Anal Bioanal Chem. 406: 4381-92. PMID: 24817354
- In Vitro Effects of Bisphenol A β-D-Glucuronide (BPA-G) on Adipogenesis in Human and Murine Preadipocytes. | Boucher, JG., et al. 2015. Environ Health Perspect. 123: 1287-93. PMID: 26018136
- In vitro assessment of the glucose-lowering effects of berberrubine-9-O-β-D-glucuronide, an active metabolite of berberrubine. | Yang, N., et al. 2017. Acta Pharmacol Sin. 38: 351-361. PMID: 28042874
- Oral Systemic Bioavailability of Bisphenol A and Bisphenol S in Pigs. | Gayrard, V., et al. 2019. Environ Health Perspect. 127: 77005. PMID: 31313948
- Different bisphenols induce non-monotonous changes in miRNA expression and LINE-1 methylation in two cell lines. | Oldenburg, J., et al. 2021. Environ Epigenet. 7: dvab011. PMID: 34858639
- Comparative metabolism of clenbuterol by rat and bovine liver microsomes and slices. | Zalko, D., et al. 1998. Drug Metab Dispos. 26: 28-35. PMID: 9443849
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bisphenol A β-D-Glucuronide, 1 mg | sc-210951 | 1 mg | $406.00 |
