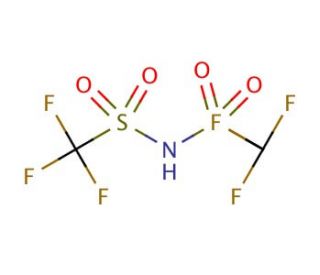
Bis(trifluoromethane)sulfonimide (CAS 82113-65-3)
See product citations (1)
QUICK LINKS
Bis(trifluoromethane)sulfonimide is a compound that functions as an electrolyte additive in battery applications and development. It plays a role in improving the performance and stability of lithium-ion batteries by enhancing the ionic conductivity of the electrolyte. Bis(Trifluoromethane)Sulfonimide facilitates the transport of lithium ions between the electrodes, leading to improved battery efficiency and cycling stability. At the molecular level, bis(trifluoromethane)sulfonimide interacts with the electrolyte components, influencing the formation of a stable solid-electrolyte interface on the electrode surface. This interaction helps to mitigate side reactions and degradation processes, thereby contributing to the performance and longevity of the battery. Bis(trifluoromethane)sulfonimide′s mechanism of action involves optimizing the electrolyte composition and enhancing the electrochemical properties of the battery system.
Bis(trifluoromethane)sulfonimide (CAS 82113-65-3) References
- High Luminescence Efficiency in MoS2 Grown by Chemical Vapor Deposition. | Amani, M., et al. 2016. ACS Nano. 10: 6535-41. PMID: 27291297
- Superacid Passivation of Crystalline Silicon Surfaces. | Bullock, J., et al. 2016. ACS Appl Mater Interfaces. 8: 24205-11. PMID: 27553365
- Modeling Insight into Battery Electrolyte Electrochemical Stability and Interfacial Structure. | Borodin, O., et al. 2017. Acc Chem Res. 50: 2886-2894. PMID: 29164857
- Near-field exciton imaging of chemically treated MoS2 monolayers. | Kim, Y., et al. 2018. Nanoscale. 10: 8851-8858. PMID: 29714393
- Incorporation of oxygen atoms as a mechanism for photoluminescence enhancement of chemically treated MoS2. | Schwermann, C., et al. 2018. Phys Chem Chem Phys. 20: 16918-16923. PMID: 29904778
- Atomic Observation of Filling Vacancies in Monolayer Transition Metal Sulfides by Chemically Sourced Sulfur Atoms. | Roy, S., et al. 2018. Nano Lett. 18: 4523-4530. PMID: 29921125
- Tuning carrier concentration in a superacid treated MoS2 monolayer. | Molas, MR., et al. 2019. Sci Rep. 9: 1989. PMID: 30760791
- Enhancing Photoluminescence and Mobilities in WS2 Monolayers with Oleic Acid Ligands. | Tanoh, AOA., et al. 2019. Nano Lett. 19: 6299-6307. PMID: 31419143
- Photoactivation of Strong Photoluminescence in Superacid-Treated Monolayer Molybdenum Disulfide. | Yamada, Y., et al. 2020. ACS Appl Mater Interfaces. 12: 36496-36504. PMID: 32635713
- Manipulating anion intercalation enables a high-voltage aqueous dual ion battery. | Huang, Z., et al. 2021. Nat Commun. 12: 3106. PMID: 34035250
- Efficiently Improved Photoluminescence in Cesium Lead Halide Perovskite Nanocrystals by Using Bis(trifluoromethane)sulfonimide. | Liu, M., et al. 2022. J Phys Chem Lett. 13: 1519-1525. PMID: 35133165
- Synergetic photoluminescence enhancement of monolayer MoS2 via surface plasmon resonance and defect repair. | Zeng, Y., et al. 2018. RSC Adv. 8: 23591-23598. PMID: 35540286
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bis(trifluoromethane)sulfonimide, 5 g | sc-252471 | 5 g | $93.00 | |||
Bis(trifluoromethane)sulfonimide, 25 g | sc-252471A | 25 g | $359.00 |
