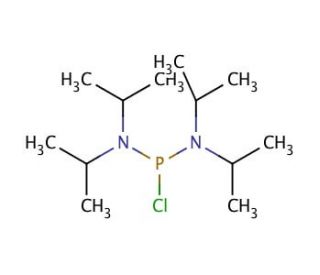
Molecular structure of Bis(diisopropylamino)chlorophosphine, CAS Number: 56183-63-2
Bis(diisopropylamino)chlorophosphine (CAS 56183-63-2)
Alternate Names:
Chlorobis(N,N-diisopropyl)phosphoramidite, Chlorobis(N,N-diisopropylamino)phosphine, Tetraisopropylphosphorodiamidous chloride
Application:
Bis(diisopropylamino)chlorophosphine is a compound containing various amounts of diisopropylamine hydrochloride
CAS Number:
56183-63-2
Purity:
≥95%
Molecular Weight:
266.79
Molecular Formula:
C12H28ClN2P
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Bis(diisopropylamino)chlorophosphine (BDIAC) contains varying amounts of diisopropylamine hydrochloride and is an organophosphorus compound used in a variety of chemical reactions. It is a useful reagent due to its ability to react with a many different substrates, producing a range of products. Common applications include organic synthesis, drug synthesis, and biochemistry. For example, Bis(diisopropylamino)chlorophosphine has been employed to synthesize alkynes, alkenes, amines, alcohols, antibiotics, anti-inflammatory agents, antifungal agents, proteins, peptides, and enzymes.
Bis(diisopropylamino)chlorophosphine (CAS 56183-63-2) References
- Adenosine 5'-O-(1-boranotriphosphate) derivatives as novel P2Y(1) receptor agonists. | Nahum, V., et al. 2002. J Med Chem. 45: 5384-96. PMID: 12431066
- 2',3'-dideoxynucleoside 5'-beta, gamma-(difluoromethylene) triphosphates with alpha-P-thio or alpha-P-seleno modifications: synthesis and their inhibition of HIV-1 reverse transcriptase. | Boyle, NA., et al. 2005. Nucleosides Nucleotides Nucleic Acids. 24: 1651-64. PMID: 16438041
- Solid-phase synthesis of symmetrical 5',5'-dinucleoside mono-, di-, tri-, and tetraphosphodiesters. | Ahmadibeni, Y. and Parang, K. 2007. Org Lett. 9: 4483-6. PMID: 17915884
- Synthesis of Site-Specifically Phosphate-Caged siRNAs. | Wu, L., et al. 2015. Curr Protoc Nucleic Acid Chem. 61: 6.12.1-6.12.15. PMID: 26344229
- Chemoselective triazole-phosphonamidate conjugates suitable for photorelease. | Siebertz, KD. and Hackenberger, CPR. 2018. Chem Commun (Camb). 54: 763-766. PMID: 29308492
- Vinylphosphonites for Staudinger-induced chemoselective peptide cyclization and functionalization. | Kasper, MA., et al. 2019. Chem Sci. 10: 6322-6329. PMID: 31341586
- Synthesis of Cell-Permeable N-Acetylhexosamine 1-Phosphates. | Kakde, BN., et al. 2021. J Org Chem. 86: 18257-18264. PMID: 34618463
- Synthesis and Anti-dengue Virus Activity of 5-Ethynylimidazole-4-carboxamide (EICA) Nucleotide Prodrugs. | Nakamura, M., et al. 2022. Chem Pharm Bull (Tokyo). 70: 220-225. PMID: 34955490
- 2-(2-Pyridyl)ethyl group: new type protecting group in the synthesis of DNA via phosphoramidite intermediates. | Hamamoto, S., et al. 1986. Nucleic Acids Symp Ser. 93-6. PMID: 3562286
- New Insight into the Reactivity of S,S-Bis-ylide. | Authesserre, U., et al. 2023. Molecules. 28: PMID: 37110528
- Synthesis of alkylphosphon(othio)ate analogues of DNA | J.C.P.F Roelen, H van den Elst, C.E Dreef, G.A van der Marel, J.H van Boom. 1992. Tetrahedron Letters. 33: 2357-2360.
- Chlorotropy in the phosphorus–carbon diad† | Oleg I. Kolodiazhnyi, Vladimir E. Grishkun. 1998. Heteroatom Chemistry. 9: 219-228.
- Novel phosphate ester-linked resins: The solid-phase generation of phenyl phosphate-containing compounds for SH2 inhibition | Chester A. Metcalf III, Chi B. Vu, Raji Sundaramoorthi, Virginia A. Jacobsen, Edgardo A. Laborde †, Jeremy Green ‡, Yinka Green §, Karina J. Macek, Taylor J. Merry, Selvi G. Pradeepan, Mayumi Uesugi, Vaibhav M. Varkhedkar, Dennis A. Holt. 1998. Tetrahedron Letters. 39: 3435-3438.
- Palladium-Catalysed Amination of Aryl- and Heteroaryl Halides Using tert-Butyl Tetraisopropylphosphorodiamidite as an Easily Accessible and Air-Stable Ligand | Gheorghe-Doru Roiban, Gerlinde Mehler, Manfred T. Reetz. 2014. European Journal of Organic Chemistry. 2014: 2070-2076.
- An improved process for the release of synthetic DNA sequences from a solid-phase capture support | Andrzej Grajkowski 1, Brian M. Cawrse 1, Mayumi Takahashi, Serge L. Beaucage. 2022. Tetrahedron Letters. 106.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bis(diisopropylamino)chlorophosphine, 5 g | sc-252457 | 5 g | $112.00 |
