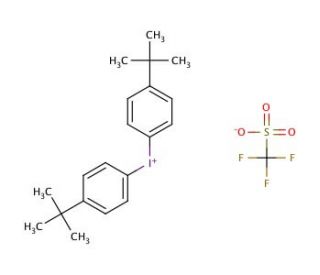
Molecular structure of Bis(4-tert-butylphenyl)iodonium triflate
Bis(4-tert-butylphenyl)iodonium triflate (CAS 84563-54-2)
Alternate Names:
Bis(4-tert-butylphenyl)iodonium trifluoromethanesulfonate; DtBPIT
Application:
Bis(4-tert-butylphenyl)iodonium triflate is a cationic photoinitiator and photoacid generator
CAS Number:
84563-54-2
Molecular Weight:
542.39
Molecular Formula:
C21H26F3IO3S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Bis(4-Tert-Butylphenyl)Iodonium Triflate acts as a powerful photoinitiator due to its ability to generate acid upon irradiation, which catalyzes the polymerization of epoxides and other cationically polymerizable monomers. Bis(4-tert-butylphenyl)iodonium triflate is significant for its role in the synthesis of complex organic molecules, where it is used to introduce iodonium groups into organic frameworks, aiding in further functionalization reactions. Bis(4-tert-butylphenyl)iodonium triflate also contributes to studies on the stability and reactivity of iodonium salts, which are key for optimizing conditions in synthetic pathways.
Bis(4-tert-butylphenyl)iodonium triflate (CAS 84563-54-2) References
- Enhanced detection of sulfo-peptides as onium salts in matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. | Ueki, M. and Yamaguchi, M. 2006. Rapid Commun Mass Spectrom. 20: 1615-20. PMID: 16636994
- Reaction of arenes with iodine in the presence of potassium peroxodisulfate in trifluoroacetic acid. Direct and simple synthesis of diaryliodonium triflates. | Hossain, MD., et al. 2006. J Org Chem. 71: 9903-5. PMID: 17168620
- Unexpected Behavior of the Heaviest Halogen Astatine in the Nucleophilic Substitution of Aryliodonium Salts. | Guérard, F., et al. 2016. Chemistry. 22: 12332-9. PMID: 27305065
- Review of Recent Inkjet-Printed Capacitive Tactile Sensors. | Salim, A. and Lim, S. 2017. Sensors (Basel). 17: PMID: 29125584
- The synthesis, characterization, and deblocking of poly(4-tert-butoxystyrene) and poly(4-tert-butoxy-.alpha.-methylstyrene) | D. A. Conlon, J. V. Crivello, J. L. Lee, and M. J. O'Brien. 1989,. Macromolecules. 22, 2,: 509–516.
- Charge transfer complexes as dual thermal/photo initiators for free-radical frontal polymerization | and Daniel P. Gary, Douglas Ngo, Amber Bui, John A. Pojman. 15 May 2022. Journal of Polymer Science. Volume60, Issue10: Pages 1624-1630.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bis(4-tert-butylphenyl)iodonium triflate, 1 g | sc-396952 | 1 g | $240.00 | |||
Bis(4-tert-butylphenyl)iodonium triflate, 5 g | sc-396952A | 5 g | $500.00 | |||
Bis(4-tert-butylphenyl)iodonium triflate, 100 g | sc-396952B | 100 g | $5000.00 | |||
Bis(4-tert-butylphenyl)iodonium triflate, 500 g | sc-396952C | 500 g | $24000.00 |
