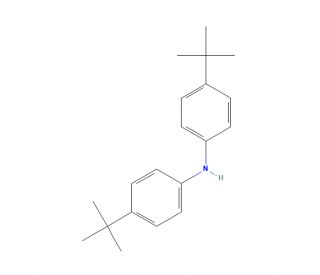
Bis(4-tert-butylphenyl)amine (CAS 4627-22-9)
QUICK LINKS
Bis(4-tert-butylphenyl)amine functions as a catalyst in organic synthesis, in the formation of carbon-carbon and carbon-heteroatom bonds. It acts as a nucleophilic catalyst, facilitating the formation of new chemical bonds through its ability to donate electron pairs. The amine group within the molecule serves as a nucleophile, allowing it to participate in various chemical reactions by forming covalent bonds with electrophilic substrates. Mode of action involves the activation of substrates through the formation of reactive intermediates, which undergo further transformations to yield the desired products. Its ability to promote bond formation and facilitate complex chemical transformations makes it a versatile in the development of new methodologies and the preparation of structurally diverse organic compounds. Bis(4-tert-butylphenyl)amine′s role in catalyzing bond-forming reactions contributes to the advancement of chemistry by enabling the efficient and selective construction of complex molecular architectures.
Bis(4-tert-butylphenyl)amine (CAS 4627-22-9) References
- Cu-Catalyzed Aerobic Oxidative N-N Coupling of Carbazoles and Diarylamines Including Selective Cross-Coupling. | Ryan, MC., et al. 2018. J Am Chem Soc. 140: 9074-9077. PMID: 29989813
- Facile Synthesis of Electroactive and Electrochromic Triptycene Poly(ether-imide)s Containing Triarylamine Units via Oxidative Electro-Coupling. | Hsiao, SH. and Liao, YC. 2017. Polymers (Basel). 9: PMID: 30965800
- Recent Advances in Heterocyclic Nanographenes and Other Polycyclic Heteroaromatic Compounds. | Borissov, A., et al. 2022. Chem Rev. 122: 565-788. PMID: 34850633
- 2,6-Dichloro-1,4-benzoquinone formation from chlorination of substituted aromatic antioxidants and its control by pre-ozonation in drinking water treatment plant. | Qiao, R., et al. 2022. Chemosphere. 299: 134498. PMID: 35390416
- Distribution and Fate of Ultraviolet Absorbents and Industrial Antioxidants in the St. Lawrence River, Quebec, Canada. | Castilloux, AD., et al. 2022. Environ Sci Technol. 56: 5009-5019. PMID: 35395156
- Push-Pull Effect of Terpyridine Substituted by Triphenylamine Motive-Impact of Viscosity, Polarity and Protonation on Molecular Optical Properties. | Maroń, AM., et al. 2022. Molecules. 27: PMID: 36296665
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bis(4-tert-butylphenyl)amine, 25 g | sc-484676 | 25 g | $39.00 |
