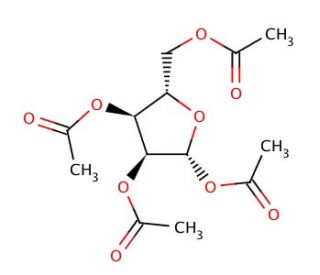
β-L-Ribofuranose 1,2,3,5-tetra-O-acetate (CAS 144490-03-9)
QUICK LINKS
β-L-Ribofuranose 1,2,3,5-tetra-O-acetate is a fully acetylated derivative of ribofuranose, utilized extensively in synthetic carbohydrate chemistry and biochemical research. The acetyl groups at the hydroxyl positions enhance the molecule′s stability and solubility in organic solvents, making it an ideal intermediate for various synthetic applications. This compound is particularly valuable in the synthesis of nucleosides and nucleotides, where the acetyl groups protect the hydroxyl functionalities during chemical reactions, allowing for selective deprotection and further modification. In research, β-L-Ribofuranose 1,2,3,5-tetra-O-acetate is used to study glycosylation mechanisms and the formation of glycosidic bonds. Its protected form allows researchers to investigate the enzymatic activities of glycosyltransferases and glycosidases, providing insights into the specificity and kinetics of these enzymes in nucleotide sugar metabolism. This compound also serves as a precursor in the synthesis of ribose-containing oligosaccharides and glycoconjugates, facilitating the exploration of carbohydrate-protein interactions and cell signaling pathways. Furthermore, β-L-Ribofuranose 1,2,3,5-tetra-O-acetate is employed in the development of glycomimetics, which mimic natural glycans and are used to probe biological functions and interactions. Its role in these synthetic and biochemical studies underscores its significance in advancing the understanding of carbohydrate chemistry and the mechanisms underlying glycosylation processes. The compound′s versatility and stability make it a valuable tool for researchers aiming to explain the complex dynamics of carbohydrate synthesis and function in biological systems.
β-L-Ribofuranose 1,2,3,5-tetra-O-acetate (CAS 144490-03-9) References
- Nucleoside Derivatives of 2,6-Diaminopurine Antivirals: Base-Modified Nucleosides with Broad-Spectrum Antimicrobial Properties. | Grazia Martina, M., et al. 2023. ChemMedChem. 18: e202300200. PMID: 37221137
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
β-L-Ribofuranose 1,2,3,5-tetra-O-acetate, 10 g | sc-257653 | 10 g | $405.00 |
