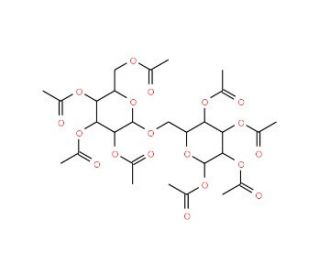
β-Gentiobiose octaacetate (CAS 4613-78-9)
QUICK LINKS
β-Gentiobiose octaacetate is a chemically modified form of β-gentiobiose, where all hydroxyl groups of the disaccharide, composed of two glucose units linked by a β-(1→6) glycosidic bond, are acetylated. This modification significantly alters its physical properties, notably enhancing its lipophilicity and decreasing its water solubility while increasing its solubility in organic solvents. In research, β-gentiobiose octaacetate is primarily utilized to study the effects of hydroxyl group protection on the stability and reactivity of glycosidic bonds in carbohydrate chemistry. It serves as an important model compound for exploring synthetic pathways and mechanisms in the assembly of complex carbohydrates and glycoconjugates. By providing insights into how acetylation affects glycosidic linkages, this compound aids scientists in developing new synthetic methods and understanding the biochemical behavior of protected sugars. Its use is crucial in advancing the field of organic synthesis, particularly in the generation of novel carbohydrate-based molecules with potential applications in various chemical and biological contexts.
β-Gentiobiose octaacetate (CAS 4613-78-9) References
- Rapid assembly of the doubly-branched pentasaccharide domain of the immunoadjuvant jujuboside A via convergent B(C6F5)3-catalyzed glycosylation of sterically-hindered precursors. | Karimov, RR., et al. 2017. Chem Commun (Camb). 53: 5838-5841. PMID: 28498382
- A new cardenolide glycoside from the roots of Streptocaulon juventas (lour.) merr. (Asclepiadaceae). | Bui, XH., et al. 2021. Nat Prod Res. 35: 1192-1198. PMID: 31368357
- Explicit-solvent molecular dynamics simulations of the β (1→ 3)-and β (1→ 6)-linked disaccharides β-laminarabiose and β-gentiobiose in water[J]. | Kony D, Damm W, Stoll S. 2004,. The Journal of Physical Chemistry B,. 108(18):: 5815-5826.
- Gentiobiosylation of β-Resorcylic Acid Esters and Lactones: First Synthesis and Characterization of Zearalenone-14-β, D-Gentiobioside[J]. | Weber J, Mikula H, Fruhmann P. 2013. Synlett,., 24(14):: 1830-1834.
- Conformational properties of glucosyl-thioglucosides and their S-oxides in solution[J]. | Sanhueza C A, Dorta R L, Vázquez J T. 2013,. Tetrahedron: Asymmetry,. 24(9-10):: 582-593.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
β-Gentiobiose octaacetate, 10 g | sc-284977 | 10 g | $500.00 | |||
β-Gentiobiose octaacetate, 25 g | sc-284977A | 25 g | $1000.00 |
