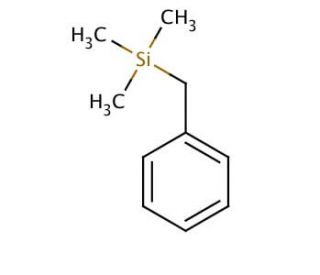
Molecular structure of Benzyltrimethylsilane, CAS Number: 770-09-2
Benzyltrimethylsilane (CAS 770-09-2)
Alternate Names:
α-Trimethylsilyltoluene
CAS Number:
770-09-2
Molecular Weight:
164.32
Molecular Formula:
C10H16Si
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Benzyltrimethylsilane is a chemical compound that functions as a protecting group in organic synthesis. It is used to temporarily mask or protect reactive functional groups in organic molecules, preventing unwanted reactions during processes. Benzyltrimethylsilane acts by forming a stable silyl ether, which can be easily removed under mild conditions, allowing the original functional group to be restored. Benzyltrimethylsilane′s mechanism of action involves the addition of the silyl group to the oxygen or nitrogen atom of the functional group, effectively blocking its reactivity.
Benzyltrimethylsilane (CAS 770-09-2) References
- Change in spin state and enhancement of redox reactivity of photoexcited states of aromatic carbonyl compounds by complexation with metal ion salts acting as Lewis acids. Lewis acid-catalyzed photoaddition of benzyltrimethylsilane and tetramethyltin via photoinduced electron transfer. | Fukuzumi, S., et al. 2001. J Am Chem Soc. 123: 7756-66. PMID: 11493049
- Diastereoselective protonation on radical anions of electron-deficient alkenes via photoinduced electron transfer. | Hayamizu, T., et al. 2004. J Org Chem. 69: 4997-5004. PMID: 15255727
- Photoinduced electron transfer reactivity of aza[60]fullerene: three discrete functionalization pathways with a single substrate. | Vougioukalakis, GC. and Orfanopoulos, M. 2004. J Am Chem Soc. 126: 15956-7. PMID: 15584717
- N-alkoxyheterocycles as irreversible photooxidants. | Wosinska, ZM., et al. 2014. Photochem Photobiol. 90: 313-28. PMID: 24354634
- α-Photooxygenation of chiral aldehydes with singlet oxygen. | Walaszek, DJ., et al. 2019. Beilstein J Org Chem. 15: 2076-2084. PMID: 31501676
- Reversal of reaction type selectivity by Lewis acid coordination: the ortho photocycloaddition of 1- and 2-naphthaldehyde. | Stegbauer, S., et al. 2019. Chem Sci. 10: 8566-8570. PMID: 31803430
- Lewis Basic Salt-Promoted Organosilane Coupling Reactions with Aromatic Electrophiles. | Reidl, TW. and Bandar, JS. 2021. J Am Chem Soc. 143: 11939-11945. PMID: 34314159
- Late-stage functionalization of 5-nitrofurans derivatives and their antibacterial activities. | Chen, G., et al. 2023. RSC Adv. 13: 3204-3209. PMID: 36756397
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyltrimethylsilane, 5 ml | sc-252430A | 5 ml | $40.00 | |||
Benzyltrimethylsilane, 25 ml | sc-252430 | 25 ml | $131.00 |
