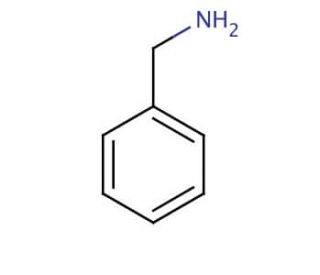
Benzylamine (CAS 100-46-9)
QUICK LINKS
Benzylamine is a colorless liquid that dissolves easily in both water and organic solvents. It holds significance as a intermediate in the synthesis of various organic compounds. Additionally, benzylamine plays a key role as an intermediate in the creation of fragrances and flavorings. Scientific research benefits greatly from the inclusion of benzylamine as a reagent. Its versatile nature enables the synthesis of diverse organic compounds, including amines, amides, and heterocyclic compounds. Moreover, benzylamine′s characteristics as a weak base allow it to function as a nucleophile in acid-base reactions and even act as a catalyst in specific reactions. When in the presence of a base, benzylamine exhibits the ability to react with an acid, resulting in the formation of an amine salt. This reaction, known as Hofmann elimination, serves as a valuable method for synthesizing various amines and amides. In essence, benzylamine′s properties and applications make it a fundamental component in both scientific research and the production of a wide array of organic substances.
Benzylamine (CAS 100-46-9) References
- Bioactivation of benzylamine to reactive intermediates in rodents: formation of glutathione, glutamate, and peptide conjugates. | Mutlib, AE., et al. 2002. Chem Res Toxicol. 15: 1190-207. PMID: 12230413
- Oxidative deamination of benzylamine by glycoxidation. | Akagawa, M., et al. 2003. Bioorg Med Chem. 11: 1411-7. PMID: 12628667
- Benzylamine exhibits insulin-like effects on glucose disposal, glucose transport, and fat cell lipolysis in rabbits and diabetic mice. | Iglesias-Osma, MC., et al. 2004. J Pharmacol Exp Ther. 309: 1020-8. PMID: 14978192
- Tyramine and benzylamine partially but selectively mimic insulin action on adipose differentiation in 3T3-L1 cells. | Subra, C., et al. 2003. J Physiol Biochem. 59: 209-16. PMID: 15000452
- Effects of oral administration of benzylamine on glucose tolerance and lipid metabolism in rats. | Bour, S., et al. 2005. J Physiol Biochem. 61: 371-9. PMID: 16180335
- Chronic benzylamine administration in the drinking water improves glucose tolerance, reduces body weight gain and circulating cholesterol in high-fat diet-fed mice. | Iffiú-Soltész, Z., et al. 2010. Pharmacol Res. 61: 355-63. PMID: 20045461
- Beta-phenylethylamine and benzylamine as substrates for human monoamine oxidase A: A source of some anomalies? | Lewinsohn, R., et al. 1980. Biochem Pharmacol. 29: 777-81. PMID: 20227955
- Derivatization of (5R)-hydroxytriptolide from benzylamine to enhance mass spectrometric detection: application to a Phase I pharmacokinetic study in humans. | Liu, J., et al. 2011. Anal Chim Acta. 689: 69-76. PMID: 21338759
- Ligand-Promoted meta-C-H Functionalization of Benzylamines. | Wang, P., et al. 2017. Angew Chem Int Ed Engl. 56: 5125-5129. PMID: 28371173
- Bioproduction of Benzylamine from Renewable Feedstocks via a Nine-Step Artificial Enzyme Cascade and Engineered Metabolic Pathways. | Zhou, Y., et al. 2018. ChemSusChem. 11: 2221-2228. PMID: 29766662
- Preparation of cytochrome P450 enzyme-cobalt phosphate hybrid nano-flowers for oxidative coupling of benzylamine. | Wu, K., et al. 2019. Enzyme Microb Technol. 131: 109386. PMID: 31615658
- Molecular Structure of Gardenia Blue Pigments by Reaction of Genipin with Benzylamine and Amino Acids. | Tsutsumiuchi, K., et al. 2021. J Agric Food Chem. 69: 3904-3911. PMID: 33761247
- Oral Supplementation with Benzylamine Delays the Onset of Diabetes in Obese and Diabetic db-/- Mice. | Iffiú-Soltesz, Z., et al. 2021. Nutrients. 13: PMID: 34444782
- Four-Step Pathway from Phenylpyruvate to Benzylamine, an Intermediate to the High-Energy Propellant CL-20. | Pandey, RP., et al. 2021. ACS Synth Biol. 10: 2187-2196. PMID: 34491727
- Enhanced Photocatalytic Coupling of Benzylamine to N-Benzylidene Benzylamine over the Organic-Inorganic Composites F70-TiO2 Based on Fullerenes Derivatives and TiO2. | Guo, Y., et al. 2023. Molecules. 28: PMID: 37298775
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzylamine, 100 ml | sc-239323 | 100 ml | $41.00 |
