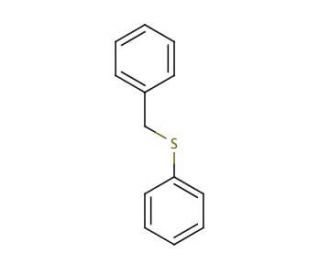
Molecular structure of Benzyl phenyl sulfide, CAS Number: 831-91-4
Benzyl phenyl sulfide (CAS 831-91-4)
Alternate Names:
Benzylthiobenzene; Benzylsulfanylbenzene
Application:
Benzyl phenyl sulfide is an aromatic sulfide
CAS Number:
831-91-4
Molecular Weight:
200.30
Molecular Formula:
C13H12S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Benzyl phenyl sulfide (BPS) is a sulfur-containing compound that belongs to the family of phenyl sulfides. In scientific research, Benzyl phenyl sulfide has found widespread use. It is employed in the synthesis of other compounds, serving as a model compound for studying the properties of other sulfides, and functioning as a reagent for the synthesis of diverse compounds. Its applications encompass the synthesis of pyridines, amides, and amines.
Benzyl phenyl sulfide (CAS 831-91-4) References
- Efficient synthesis of 1-sulfonyl-1,2,3-triazoles. | Raushel, J. and Fokin, VV. 2010. Org Lett. 12: 4952-5. PMID: 20931987
- Inhibitory effects on microbial growth using the derivatives of benzyl phenyl sulfide. | Hatano, A., et al. 2011. Biocontrol Sci. 16: 63-7. PMID: 21719991
- Palladium-Catalyzed Direct α-Aryation of Benzyl Thioethers with Aryl Bromides. | Frensch, G., et al. 2014. Adv Synth Catal. 356: 2517-2524. PMID: 25685127
- Carbon Nanotubes Modified by Venturello Complex as Highly Efficient Catalysts for Alkene and Thioethers Oxidation With Hydrogen Peroxide. | Evtushok, VY., et al. 2019. Front Chem. 7: 858. PMID: 31921779
- Efficient Photooxidation of Sulfides with Amidated Alloxazines as Heavy-atom-free Photosensitizers. | Guo, H., et al. 2020. ACS Omega. 5: 10586-10595. PMID: 32426617
- Catalytic Chemoselective Sulfimidation with an Electrophilic [CoIII (TAML)]- -Nitrene Radical Complex*. | van Leest, NP., et al. 2021. Chemistry. 27: 371-378. PMID: 32810326
- The Exchange of Cyclometalated Ligands. | Ryabov, AD. 2021. Molecules. 26: PMID: 33401624
- Ni-Catalyzed Borylation of Aryl Sulfoxides. | Huang, M., et al. 2021. Chemistry. 27: 8149-8158. PMID: 33851475
- Gold Clusters: From the Dispute on a Gold Chair to the Golden Future of Nanostructures. | Ganadu, ML., et al. 2021. Molecules. 26: PMID: 34443602
- Fe3O4@BNPs@SiO2-SO3H as a highly chemoselective heterogeneous magnetic nanocatalyst for the oxidation of sulfides to sulfoxides or sulfones. | Ghanbari Kermanshahi, M. and Bahrami, K. 2019. RSC Adv. 9: 36103-36112. PMID: 35540572
- A Green Route to Benzyl Phenyl Sulfide from Thioanisole and Benzyl Alcohol over Dual Functional Ionic Liquids. | Wu, F., et al. 2023. Chem Asian J. 18: e202201078. PMID: 36445934
- Evolution of enzyme functionality in the flavin-containing monooxygenases. | Bailleul, G., et al. 2023. Nat Commun. 14: 1042. PMID: 36823138
- Oxidative cleavage and ammoxidation of organosulfur compounds via synergistic Co-Nx sites and Co nanoparticles catalysis. | Luo, H., et al. 2023. Nat Commun. 14: 2981. PMID: 37221164
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl phenyl sulfide, 5 g | sc-233995 | 5 g | $55.00 |
