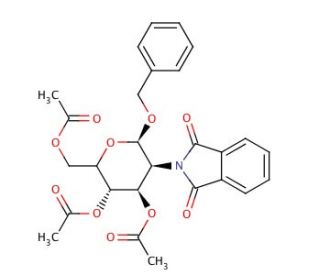
Benzyl 2-Deoxy-2-phthalimido-3,4,6-tri-O-acetyl-β-D-glucopyranoside (CAS 80035-31-0)
QUICK LINKS
Benzyl 2-Deoxy-2-phthalimido-3,4,6-tri-O-acetyl-β-D-glucopyranoside plays a pivotal role in carbohydrate chemistry research due to its strategic arrangement of protecting groups, enabling precise control over glycosylation reactions. With phthalimido, benzyl, and acetyl groups strategically positioned on the glucopyranoside ring, this compound serves as a versatile building block for the synthesis of complex oligosaccharides. The phthalimido group, selectively removable under mild conditions, exposes a free amine for further functionalization or conjugation, facilitating the synthesis of diverse glycoconjugates. Additionally, the benzyl and acetyl groups provide steric hindrance and regioselectivity during glycosylation, ensuring the formation of desired glycosidic linkages. This compound has found extensive use in the synthesis of biologically relevant glycans, including glycoproteins and glycolipids, enabling the investigation of structure-function relationships in glycobiology. Moreover, its synthetic versatility and precise control over glycosylation make it indispensable for the development of carbohydrate-based materials, such as glycomaterials and glycodendrimers, with applications in biomaterials science and bioorganic chemistry. Its utilization continues to expand in research aimed at understanding the roles of carbohydrates in biological systems and designing carbohydrate-based probes.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2-Deoxy-2-phthalimido-3,4,6-tri-O-acetyl-β-D-glucopyranoside, 500 mg | sc-207338 | 500 mg | $300.00 |
