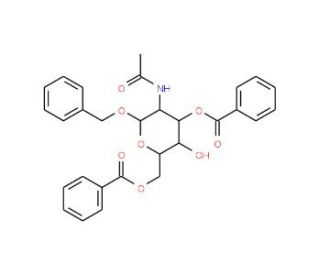

Benzyl 2-Acetamido-3,6-di-O-benzoyl-2-deoxy-α-D-glucopyranoside (CAS 82827-77-8)
QUICK LINKS
Benzyl 2-Acetamido-3,6-di-O-benzoyl-2-deoxy-α-D-glucopyranoside is a chemical compound with notable research applications, particularly in the field of carbohydrate chemistry and glycosylation studies. This compound has been utilized as a substrate or glycosyl donor in the enzymatic synthesis of complex oligosaccharides and glycoconjugates. Its specific structure, with multiple benzoyl and benzyl groups, makes it suitable for investigating stereochemical preferences and regioselectivity in glycosylation reactions catalyzed by glycosyltransferases. Researchers have employed Benzyl 2-Acetamido-3,6-di-O-benzoyl-2-deoxy-α-D-glucopyranoside as a model compound to explore the mechanistic details of glycosylation reactions and the factors influencing glycosidic bond formation, such as solvent effects, temperature, and catalyst concentration. Moreover, this compound has been used in the synthesis of glycosylated molecules with potential biological activities, including glycolipids, glycopeptides, and glycosylated natural products. Its role as a versatile glycosylation building block has contributed to advancements in carbohydrate-based drug discovery, biomaterials engineering, and chemical biology research. Overall, Benzyl 2-Acetamido-3,6-di-O-benzoyl-2-deoxy-α-D-glucopyranoside serves as a valuable tool in elucidating glycosylation mechanisms and designing bioactive compounds.
Benzyl 2-Acetamido-3,6-di-O-benzoyl-2-deoxy-α-D-glucopyranoside (CAS 82827-77-8) References
- First Synthesis of beta-D-Galf(1-4)GlcNAc, a Structural Unit Attached O-Glycosidically in Glycoproteins of Trypanosoma cruzi. | Gallo-Rodriguez, C., et al. 1996. J Org Chem. 61: 1886-1889. PMID: 11667072
- A comparative study of the influence of some protecting groups on the reactivity of D-glucosamine acceptors with a galactofuranosyl donor. | Bohn, ML., et al. 2006. Carbohydr Res. 341: 1096-104. PMID: 16630599
- Synthesis of β-d-Galp-(1→ 3)-β-d-Galp-(1→ 6)-[β-d-Galf-(1→ 4)]-d-GlcNAc, a tetrasaccharide component of mucins of Trypanosoma cruzi | Gallo-Rodriguez, C., Gil-Libarona, M. A., Mendoza, V. M., & de Lederkremer, R. M. 2002. Tetrahedron. 58(46): 9373-9380.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2-Acetamido-3,6-di-O-benzoyl-2-deoxy-α-D-glucopyranoside, 500 mg | sc-221312 | 500 mg | $360.00 |
