
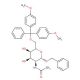
Benzyl 2-Acetamido-2-deoxy-6-dimethoxytrityl-α-D-galactopyranoside
QUICK LINKS
Benzyl 2-Acetamido-2-deoxy-6-dimethoxytrityl-α-D-galactopyranoside plays a pivotal role in carbohydrate chemistry research, primarily serving as a building block for the synthesis of complex oligosaccharides and glycoconjugates. Its mechanism of action is rooted in its structural features, which enable it to act as a glycosyl donor in glycosylation reactions. This compound is particularly valuable in the synthesis of galactose-containing oligosaccharides due to the presence of the α-D-galactopyranoside moiety. Researchers utilize Benzyl 2-Acetamido-2-deoxy-6-dimethoxytrityl-α-D-galactopyranoside to explore various aspects of carbohydrate-mediated interactions, including cell-cell recognition, host-pathogen interactions, and immune responses. By manipulating the protecting groups, such as the dimethoxytrityl and acetamido groups, scientists can precisely control the regioselectivity and stereochemistry of glycosylation reactions, enabling the synthesis of diverse glycan structures. Furthermore, this compound contributes to the investigation of glycan function in biological systems, shedding light on their roles in diverse biological processes. Its synthetic versatility and utility make it an indispensable tool for elucidating the complex roles of carbohydrates in biology and advancing our understanding of glycobiology.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2-Acetamido-2-deoxy-6-dimethoxytrityl-α-D-galactopyranoside, 50 mg | sc-221302 | 50 mg | $300.00 | |||
Benzyl 2-Acetamido-2-deoxy-6-dimethoxytrityl-α-D-galactopyranoside, 500 mg | sc-221302A | 500 mg | $1360.00 |
