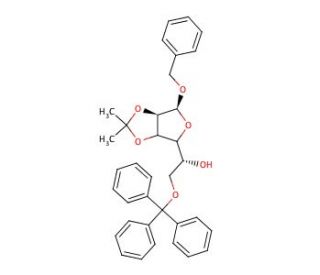
Benzyl 2,3-O-Isopropylidene-6-O-trityl-α-D-mannofuranose (CAS 91364-11-3)
QUICK LINKS
Benzyl 2,3-O-Isopropylidene-6-O-trityl-α-D-mannofuranose, a carbohydrate derivative, has garnered significant attention in scientific research for its versatile applications in carbohydrate chemistry and glycosylation reactions. Its unique structural features, including the benzyl and trityl protecting groups, make it a valuable building block for the synthesis of complex carbohydrates and glycoconjugates. The chemical exhibits a specific mechanism of action in glycosylation reactions, serving as both a glycosyl donor and acceptor, facilitating the formation of glycosidic bonds with high efficiency and selectivity. Researchers have utilized this compound extensively in the synthesis of oligosaccharides, glycopeptides, and glycolipids for various research applications, including studies on carbohydrate-protein interactions, glycan analysis, and the development of carbohydrate-based materials. Furthermore, the orthogonal reactivity provided by its protecting groups allows for selective deprotection and further functionalization, enabling the creation of diverse carbohydrate structures with tailored properties. Overall, Benzyl 2,3-O-Isopropylidene-6-O-trityl-α-D-mannofuranose remains a crucial tool in carbohydrate chemistry research, offering opportunities for the synthesis and exploration of complex glycoconjugates with implications in various scientific disciplines.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2,3-O-Isopropylidene-6-O-trityl-α-D-mannofuranose, 50 mg | sc-221324 | 50 mg | $330.00 |
