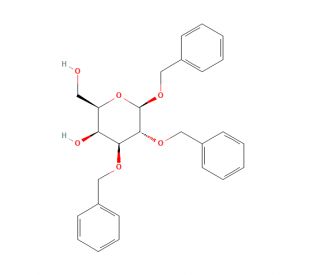
Benzyl 2,3-Di-O-benzyl-β-D-galactopyranoside (CAS 74801-06-2)
QUICK LINKS
Benzyl 2,3-Di-O-benzyl-β-D-galactopyranoside plays a significant role in carbohydrate chemistry research due to its utility as a versatile building block for synthesizing complex carbohydrates and glycoconjugates. Its structure comprises a galactopyranoside backbone with two hydroxyl groups substituted with benzyl moieties, providing accessibility for further chemical modifications. Researchers utilize this compound as a key precursor in the synthesis of various carbohydrate derivatives, including glycosides, glycopeptides, and glycolipids, through glycosylation reactions. Additionally, Benzyl 2,3-Di-O-benzyl-β-D-galactopyranoside serves as a substrate for enzymatic glycosylation processes, enabling investigations into the substrate specificity and catalytic mechanisms of glycosyltransferases. Furthermore, its structural features make it suitable for studying carbohydrate-protein interactions and carbohydrate-mediated biological recognition events. By employing this compound, researchers can explain the roles of carbohydrates in cellular processes, host-pathogen interactions, and immune responses. Overall, Benzyl 2,3-Di-O-benzyl-β-D-galactopyranoside serves as a valuable tool in carbohydrate chemistry research, facilitating the synthesis of diverse carbohydrate derivatives and contributing to a deeper understanding of carbohydrate-related biological phenomena.
Benzyl 2,3-Di-O-benzyl-β-D-galactopyranoside (CAS 74801-06-2) References
- The intermolecular migration of polyol stannylenes as a reaction contributing to the regioselectivity of substitution. | David, S. and Malleron, A. 2000. Carbohydr Res. 329: 215-8. PMID: 11086702
- Anchimeric assistance by the anomeric phenylthio group in the nucleophilic substitution of a 6-O-trifluoromethanesulfonyl-β-d-galactopyranoside | Compain-Batissou, M., Mesrari, L., Anker, D., & Doutheau, A. 1999. Carbohydrate research. 316(1-4): 201-205.
- A strategy for chemical synthesis of selectively methyl-esterified oligomers of galacturonic acid | Clausen, M. H., Jørgensen, M. R., Thorsen, J., & Madsen, R. 2001. Journal of the Chemical Society, Perkin Transactions 1. 5: 543-551.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2,3-Di-O-benzyl-β-D-galactopyranoside, 25 mg | sc-221319 | 25 mg | $300.00 |
