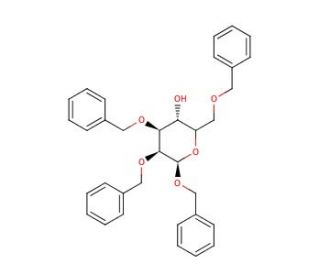
Benzyl 2,3,6-Tri-O-benzyl-β-D-glucopyranoside (CAS 67831-42-9)
QUICK LINKS
Benzyl 2,3,6-tri-O-benzyl-β-D-glucopyranoside, a derivative of glucose, has garnered significant interest in scientific research, particularly in carbohydrate chemistry and chemical biology. Its mechanism of action primarily revolves around its role as a glycosylation agent and a building block in the synthesis of complex carbohydrates and glycoconjugates. Chemists utilize this compound as a glycosyl donor in glycosylation reactions, enabling the regioselective introduction of glucose residues onto various acceptor molecules. Furthermore, its tri-O-benzyl protection provides enhanced stability and reactivity, allowing for efficient glycosylation under mild conditions. Researchers have employed Benzyl 2,3,6-tri-O-benzyl-β-D-glucopyranoside in the synthesis of oligosaccharides, polysaccharides, glycolipids, and glycoproteins with tailored structures and properties. Its versatile reactivity and compatibility with various glycosylation methodologies make it a valuable tool for constructing structurally diverse carbohydrates and probing their biological functions. Additionally, this compound serves as a precursor for the preparation of carbohydrate-based probes and biomaterials for investigating carbohydrate-protein interactions, cell recognition processes, and glycobiology. Its significance in carbohydrate chemistry research underscores its utility in studying the roles of carbohydrates in biological systems and designing novel biomolecules with potential applications in biotechnology, diagnostics, and materials science.
Benzyl 2,3,6-Tri-O-benzyl-β-D-glucopyranoside (CAS 67831-42-9) References
- Total synthesis of the mollu-series glycosyl ceramides alpha-D-Manp-(1----3)-beta-D-Manp-(1----4)-beta-D-Glcp-(1----1)-Cer and alpha-D-Manp-(1----3)-[beta-D-Xylp-(1----2)]-beta-D-Manp-(1----4)-beta- D-Glcp-(1----1)-Cer. | Mori, M., et al. 1990. Carbohydr Res. 195: 199-224. PMID: 2331702
- Synthesis of cellobiose, cellotriose, cellotetraose, and lactose | Okushio, K., Fukuyama, K., & Kuge, T. 1983. Carbohydrate research,. 121: 163-173.
- 1, 6-Epithio-and 1, 6-Episeleno-β-D-glucopyranose: Useful Adjuncts in the Synthesis of 6-Deoxy-β-D-glucopyranosides | Stick, R. V., Tilbrook, D. M. G., & Williams, S. J. 1999. Australian journal of chemistry. 52(7): 685-694.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2,3,6-Tri-O-benzyl-β-D-glucopyranoside, 50 mg | sc-221331 | 50 mg | $380.00 |
